Evidence Is the Product – Rethinking U.S FDA Device Testing
March 5, 2026
Navigating FDA expectations doesn’t have to be a guessing game.
-
- Does the device perform as described?
- Have relevant risks been identified and mitigated?
- Is patient interaction acceptably safe?
- Does evidence support the intended use and labeling?
- Where necessary, are clinical data sufficient to address residual questions?
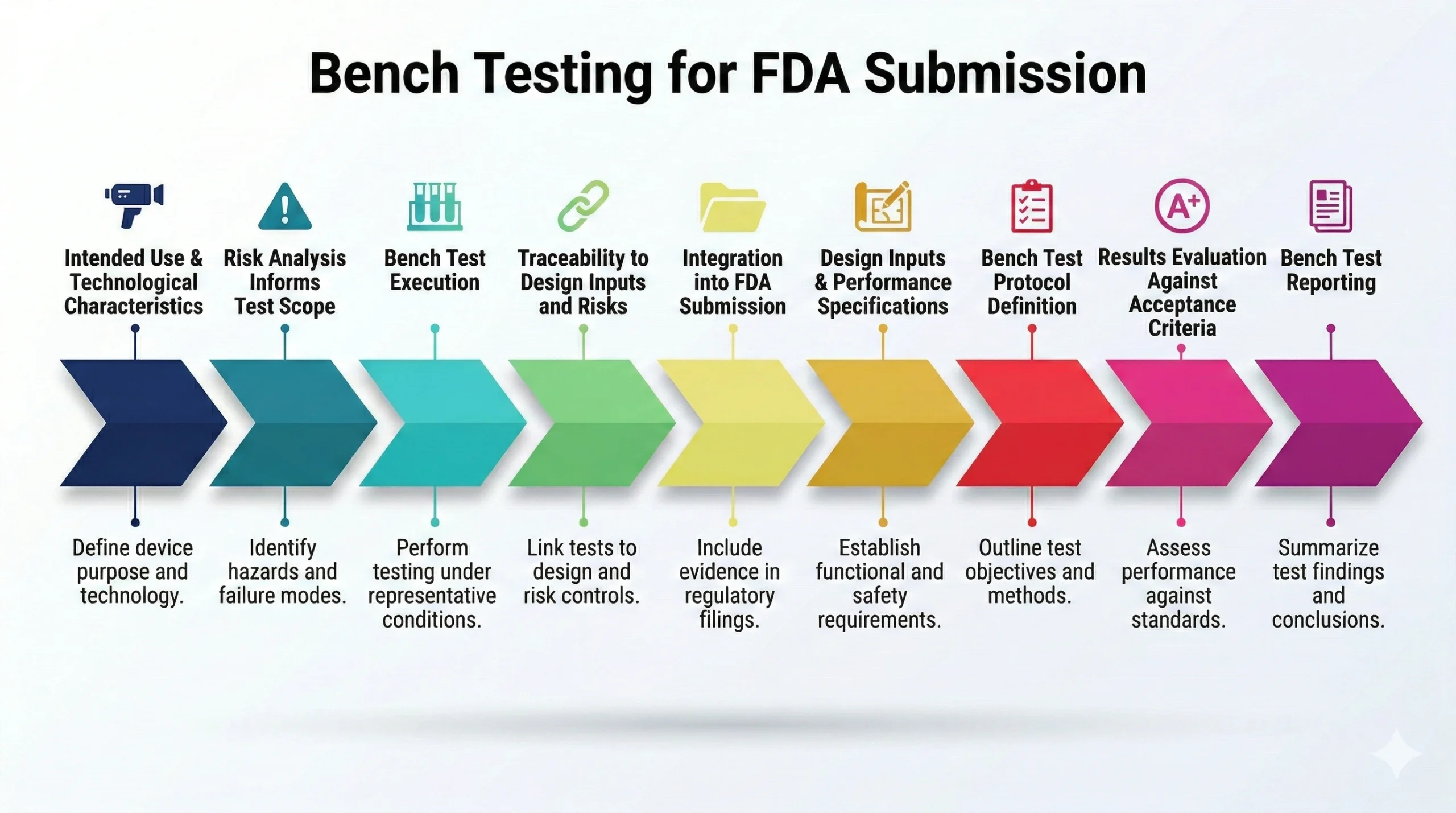
Across 510(k), De Novo, and PMA pathways, the FDA applies a clear, science‑driven logic focused on performance, risk mitigation, patient safety, and intended use. Because evidence requirements scale with risk and technological novelty, testing may involve anything from basic verification to extensive bench, biocompatibility, software, or clinical studies. This edition breaks down that logic into actionable insight for medtech sponsors.
Bench Testing: Foundational Evidence
Bench testing translates design intent into objective evidence. It is the core source of nonclinical performance data, demonstrating that devices meet specifications and perform consistently under anticipated conditions. Bench testing verifies that design outputs conform to inputs and that risk controls are supported by measurable criteria.
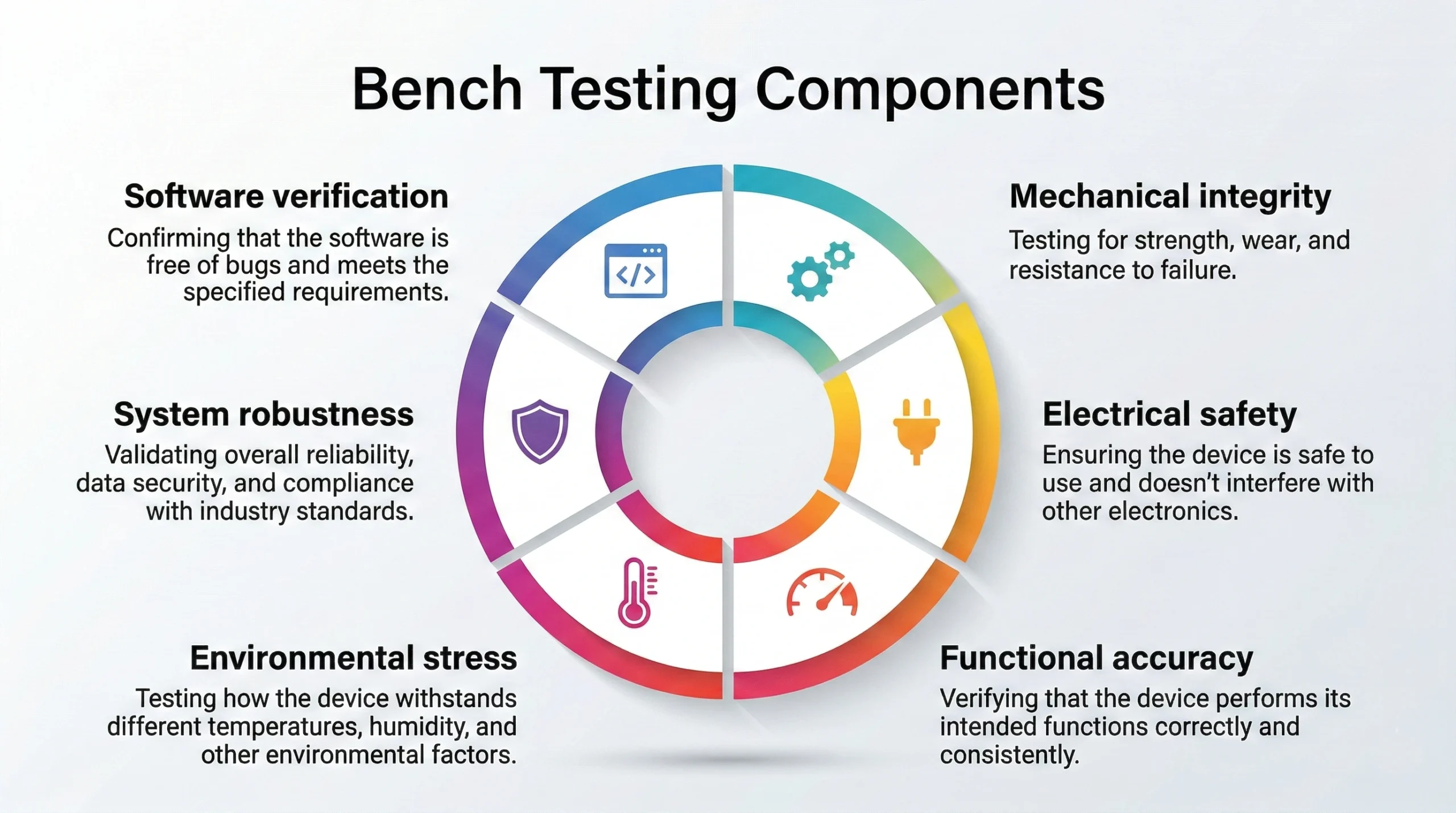
FDA guidance on nonclinical bench performance testing requires predefined protocols, acceptance criteria, and traceable results. Review emphasizes scientific rationale, methodological rigor, and completeness. Testing should directly support performance claims or risk mitigation, with scope determined by intended use, technological characteristics, and device risk.
When Code Is the Device: Software Verification and Validation for SaMD and AI-Enabled Technologies
For devices incorporating software, verification and validation are central to bench testing. The FDA evaluates requirements for traceability, risk analysis, verification testing, validation against intended use, and configuration control. For embedded software, these activities support functional and safety claims.
Software as a Medical Device (SaMD) relies on software validation to demonstrate that intended outputs are reliable and safe. AI- or machine learning–enabled devices also require algorithm validation with defined training and test datasets, along with performance metrics such as sensitivity and specificity. Submissions must describe dataset characteristics, validation methods, and performance consistency relative to intended use. Bench testing, in this context, extends beyond code verification to structured evaluation of software-driven outputs.
Sterilization and Reprocessing Validation
Sterile devices must demonstrate consistent achievement of the claimed sterility assurance level, with residual sterilants within acceptable limits. Reusable devices require validated cleaning and reprocessing instructions. These studies are critical for patient safety and receive careful FDA scrutiny.
Packaging, Stability, and Shelf Life
Shelf life and packaging integrity are regulatory claims. Evidence must show the device maintains safety and performance during storage and distribution, including:
-
- Packaging integrity and seal strength testing
- Accelerated and real-time aging
- Transportation simulation
- Stability testing
Claims cannot be retroactively supported without prospective data, making early planning essential.
Biological Safety and Material Evaluation: Risk Context
Biocompatibility testing assesses whether device materials that contact the body may cause unacceptable adverse biological responses. FDA emphasizes a risk-based approach, including:
-
- Material composition and processing evaluation
- Consideration of contact type and duration
- Identification of relevant biological endpoints
- Scientific justification for testing decisions
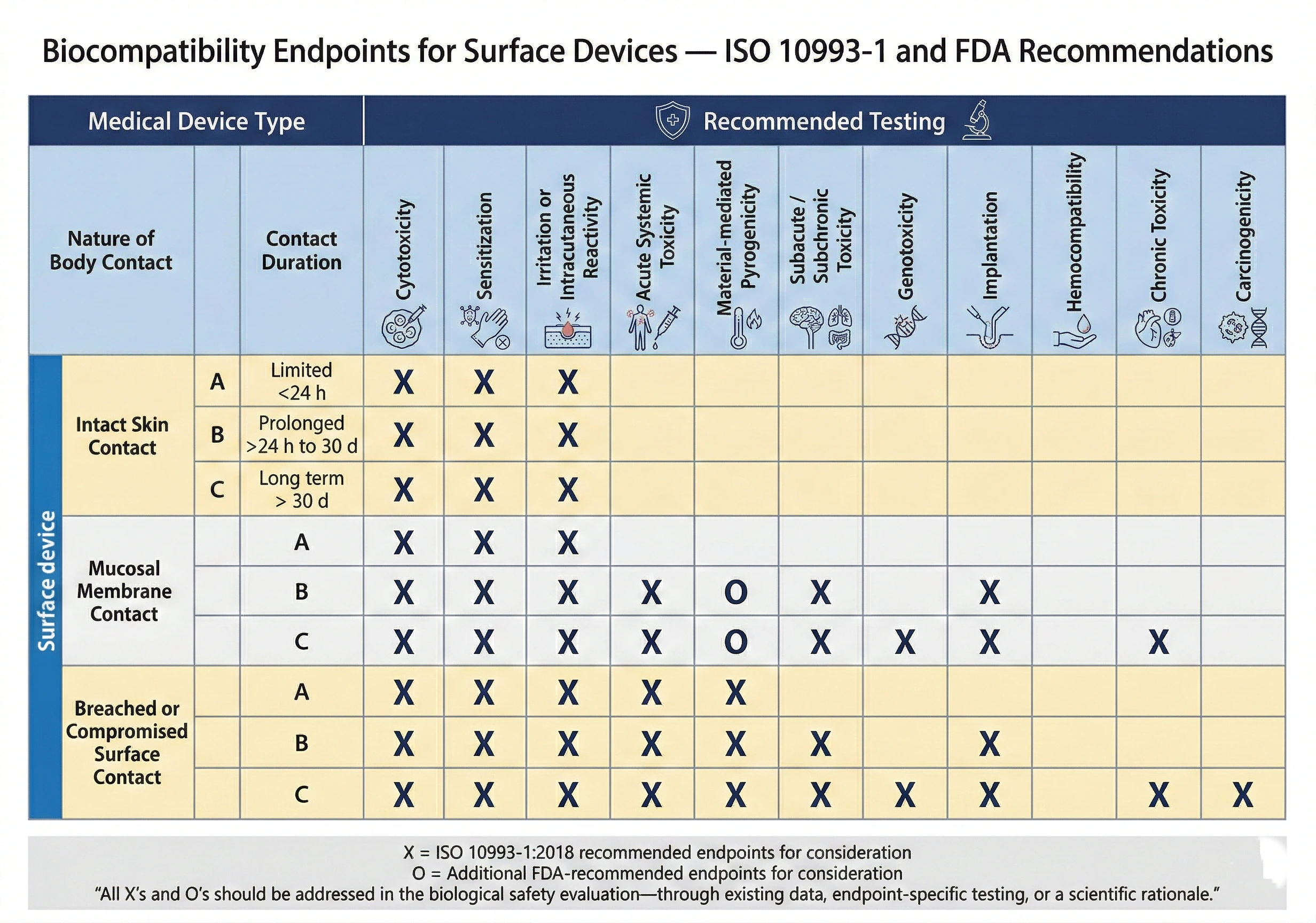
Evaluation applies to devices with direct or indirect patient contact. Modifications that do not affect tissue-contacting components may not require additional testing, but any change that could indirectly affect patient-contacting materials warrants reassessment. ISO 10993-1 endpoints guide testing selection, with additional FDA-recommended endpoints incorporated as appropriate.
The figure below summarizes the ISO 10993‑1 endpoints recommended by the FDA for surface devices that contact the user’s skin:
Nonclinical and Clinical Studies: You Can’t Bench-Test Biology
When bench testing cannot fully address safety or performance in a biological environment, additional nonclinical studies are required. These studies assess physiological interactions, such as implant stability, degradation, thrombogenicity, energy delivery effects, or procedural feasibility. Animal models are used to answer defined safety or functional questions, not to replicate human trials, reducing uncertainty before first-in-human use.
Clinical investigations are necessary when nonclinical evidence is insufficient or when novel safety or effectiveness questions arise. FDA evaluates study design, endpoint selection, statistical methods, and data integrity. Clinical endpoints must be clinically meaningful and aligned with labeling claims. Together, nonclinical and clinical studies complete the evidence package for regulatory review.
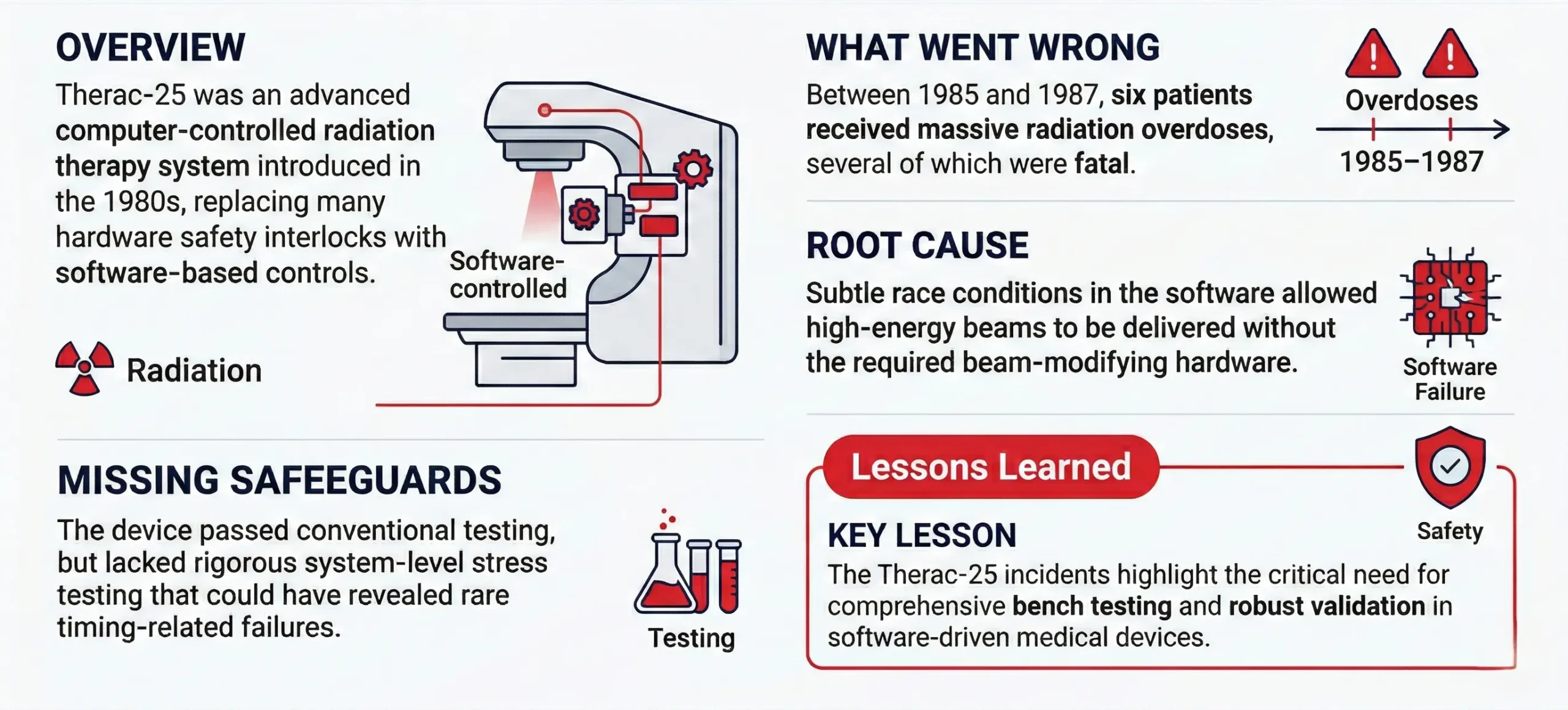
Case study: The Therac-25 Tragedy
Medical Device Recall Trends
In 2024, U.S. medical device recalls reached a four-year high, with 1,059 events recorded, up 8.6% from 975 in 2023 (Sedgwick, 2025). Units affected rose from ~283 million to over 440 million. Device failure led recall causes, with quality and software issues remaining prominent.
Early 2025 data show that total recall events and impacted units decreased in Q1, but Class I recalls, the FDA’s most serious recall category, indicating a reasonable probability of serious adverse health consequences or death, increased compared to the previous quarter. These trends highlight the ongoing importance of rigorous premarket evidence planning and post-market vigilance.
Key takeaways
-
- Classify early and accurately—classification guides testing expectations and regulatory pathway.
- Iterative testing—early bench and hazard evaluation reduces late-stage redesign risk.
- Leverage FDA-recognized standards—align tests with ISO and FDA consensus standards.
- Plan software validation comprehensively—integrate hazard analysis early.
- Document everything with traceability—reviewers must see the logic from risk to evidence to conclusion.
Resource corner
Insights from the MedTech Guru
Q1: How much bench testing is enough before clinical trials?
A: Relevance matters more than quantity. Evidence must identify and mitigate hazards with traceability to design inputs. Over-testing wastes resources and under-testing exposes regulatory risk.
Q2: Can software validation alone replace bench tests in SaMD or AI devices?
A: No, end-to-end evidence is expected. Verification, model validation, and performance testing must collectively demonstrate that device outputs meet specifications under real-world conditions.
Q3: How do you test devices with multiple interacting subsystems?
A: Integration and system-level testing are essential. Subsystem tests alone can miss emergent failure modes.
Q4: How do you validate AI/ML training datasets for submission?
A: Traceability, reproducibility, and bias mitigation are key. Documentation must link datasets, training, and validation to performance claims.
Upcoming Events
Advanced Healthcare 2026. Apr 1–2, 2026, Berlin, Germany.
Health 2.0 Conference. Apr 7–9, 2026, Las Vegas, NV, USA
Innovation Summit Dublin. Apr 28–30, 2026, The Shelbourne Dublin, Dublin, Ireland
How Can BLA Regulatory Help?
BLA Regulatory, LLC operates as a global regulatory consulting firm specializing in medical device and biopharmaceutical compliance and submission support. We help medtech sponsors shape risk-based evidence strategies, advise on applicable testing and regulatory expectations, and prepare coherent submissions that integrate test outcomes with safety and performance claims. With deep experience in U.S. FDA pathways, including IND, NDA, BLA, 510(k), PMA, De Novo, and Breakthrough Device designations, the firm supports sponsors through active agency interactions. BLA Regulatory serves clients across the U.S., Europe, China, and Japan, helping bring safe and effective innovations to market with speed and reliability. For more insights, visit: https://bla-regulatory.com/
Stay in Orbit
Subscribe to future issues of MedTech Orbit for practical updates, U.S. FDA device regulations, and smarter compliance strategies, delivered straight to your inbox.

