Why US FDA Demands Human Factors Engineering for Devices?
April 2, 2026
In the MedTech industry, innovation often centers on what a device can do, such as detect disease earlier, or deliver therapy more precisely. Yet from a regulatory standpoint, the most critical question is often simpler: can people use the device safely and correctly in the real world?
Many device-related adverse events are not caused by mechanical failures or software defects. Instead, they arise from use-related errors, situations where the interaction between the user and the device breaks down. A medical device is only as safe and effective as the human being’s ability to use it correctly.
Human Factors Engineering (HFE), also known as Usability Engineering (UE), addresses this gap and focuses on designing medical devices that align with human capabilities, limitations, and real-world operating conditions.
Understanding Human–Device Interaction
The user interface represents the central interaction zone between the user and the device. It includes all components with which users interact during activities such as preparing the device for use (unpacking, setup, calibration), operating the device, monitoring therapy or diagnostic results, maintaining the device (cleaning, replacing batteries, servicing) etc.
In other words, every interaction point between the user and the device is part of the interface. From a regulatory perspective, device manufacturers must demonstrate that this interface has been intentionally designed to minimize the likelihood of user error.
Below, we break down the Device User Interface in Operational Context:
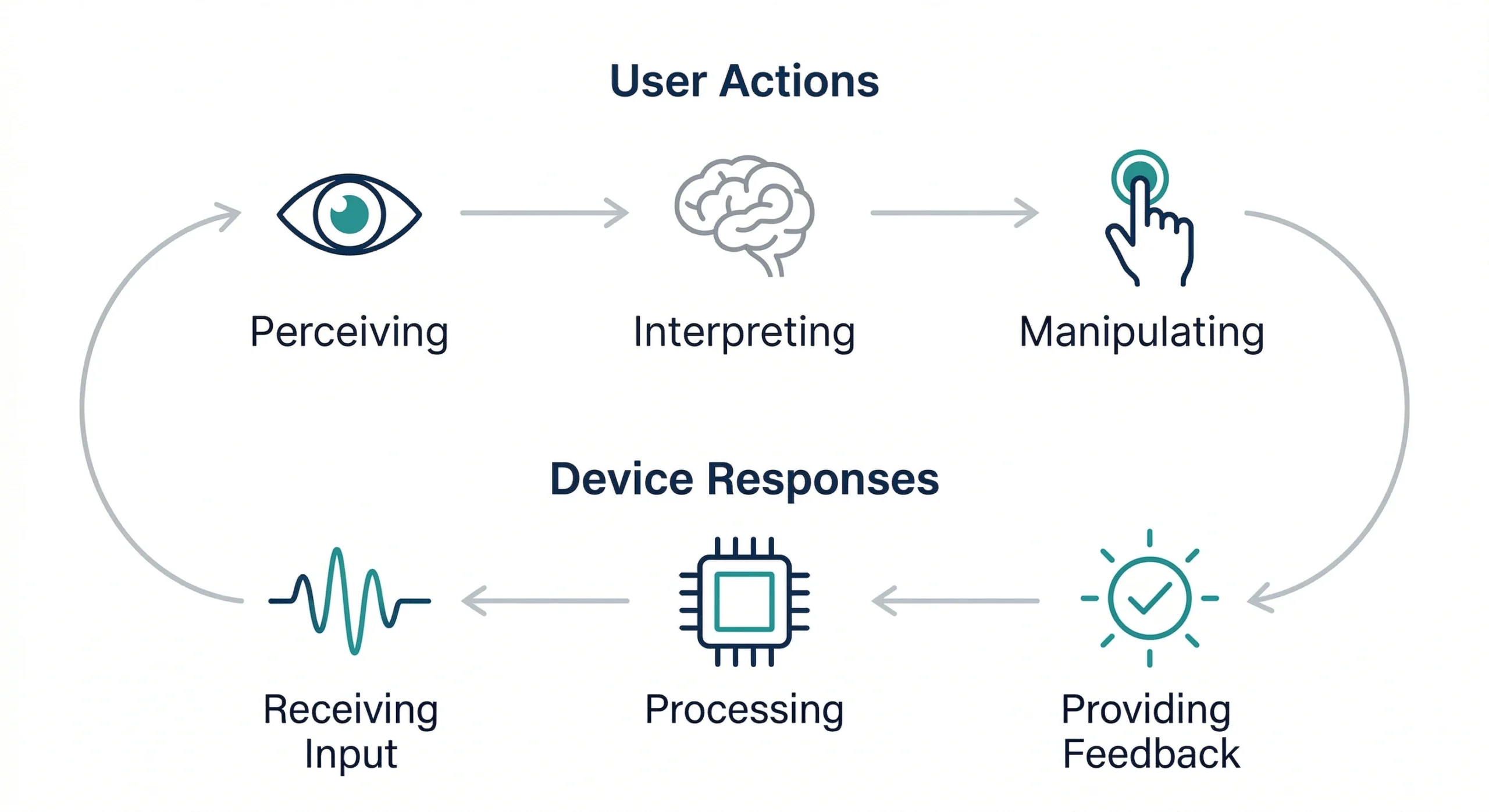
Source: Adapted from Redmill and Rajan, 1997
How Users Interact with Devices
Human factors analysis examines how people perform three fundamental actions when using medical devices:
-
- Perceiving Information: Users receive information from the device through displays, indicators, alarms, labels, and tactile cues. For example, visual displays showing vital signs, audio alarms indicating abnormal conditions, and indicator lights signalling device status etc. The clarity and visibility of this information directly affect user performance.
- Interpreting Information and Making Decisions: Once information is perceived, users must interpret it and decide what action to take. This cognitive step is influenced by time pressure, stress levels, training, and environmental distractions. Poorly designed interfaces can lead to misinterpretation, which may result in incorrect actions.
- Manipulating the Device: Users then interact with the device by performing actions such as adjusting settings, replacing components, initiating or stopping therapy, connecting accessories, or performing maintenance. These actions must be intuitive and physically manageable for the intended user population.
How Devices Respond to Users
Interaction is not one-sided. Devices also participate in the communication process. Medical devices must:
-
- Receive input from the user, such as a button press or configuration change
- Process the input and execute the requested function
- Provide feedback indicating the result of the action
Effective feedback mechanisms include confirmation screens, audible alerts, status indicators, or display updates. Without clear feedback, users may assume an action occurred when it did not, potentially leading to hazardous situations.
Why Human Factors Engineering Matters for Medical Devices

The Human Factors Engineering Lifecycle
From a regulatory perspective, human factors engineering should be integrated throughout device development.
Use-Related Risk Analysis
This stage identifies potential user errors and evaluates their potential impact on patient safety. Common sources of risk include:
-
- Incorrect device setup
- Misinterpretation of displays
- Incorrect parameter entry
- Failure to respond appropriately to alarms
These risks are typically evaluated within the broader ISO 14971 risk management framework.
Formative Usability Testing
Formative testing occurs during development and helps refine the design through early user feedback. These studies may include:
-
- Task simulations
- Prototype evaluations
- Workflow observations
The goal is to identify usability issues before final design decisions are locked in.
Summative (Validation) Testing
Human factors validation studies occur near the end of development. In these studies:
-
- Representative users perform critical tasks
- Testing occurs in simulated real-world environments
- Participants receive only the training expected in real-world use
The results demonstrate whether the device can be used safely and effectively without additional assistance.
Case Studies
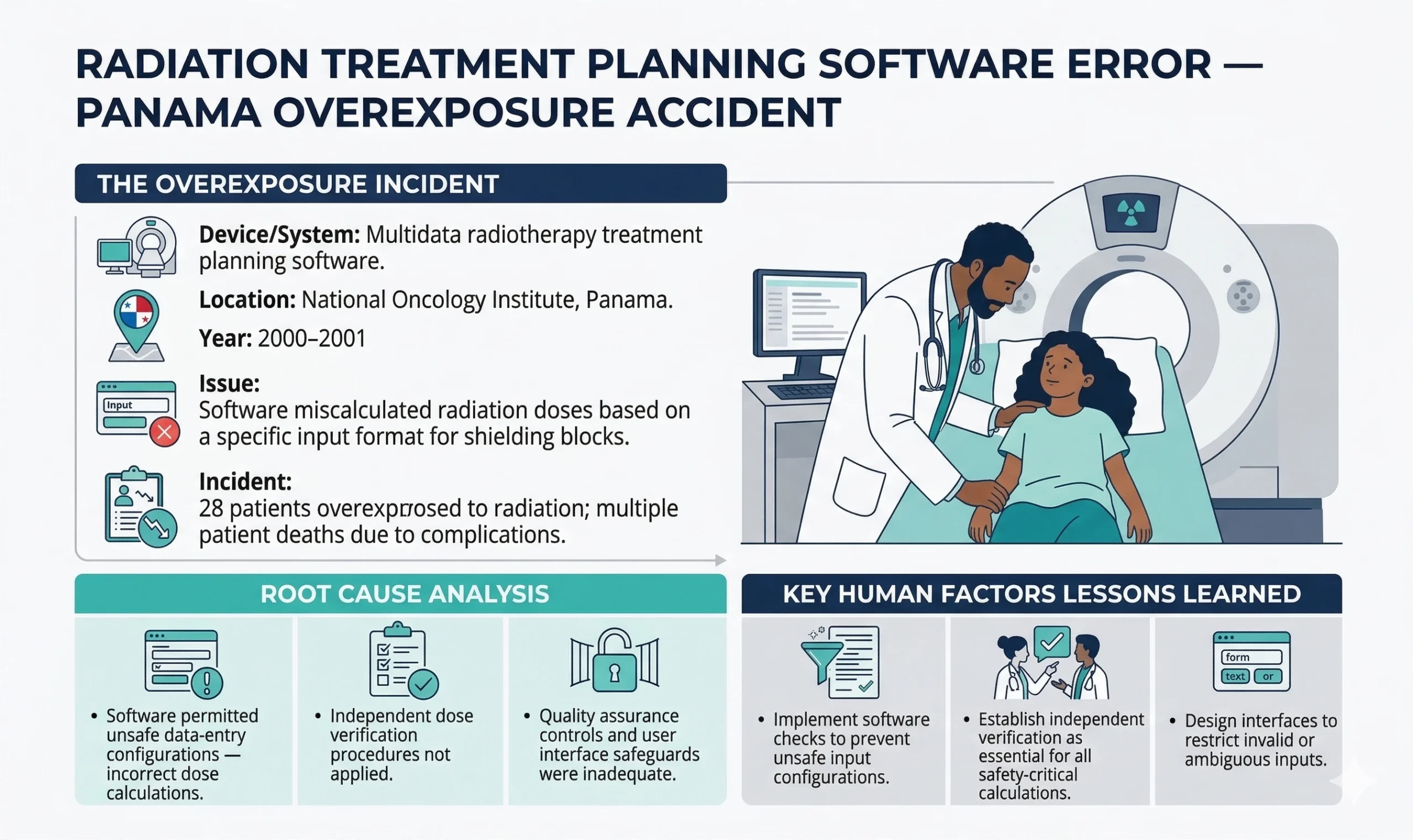
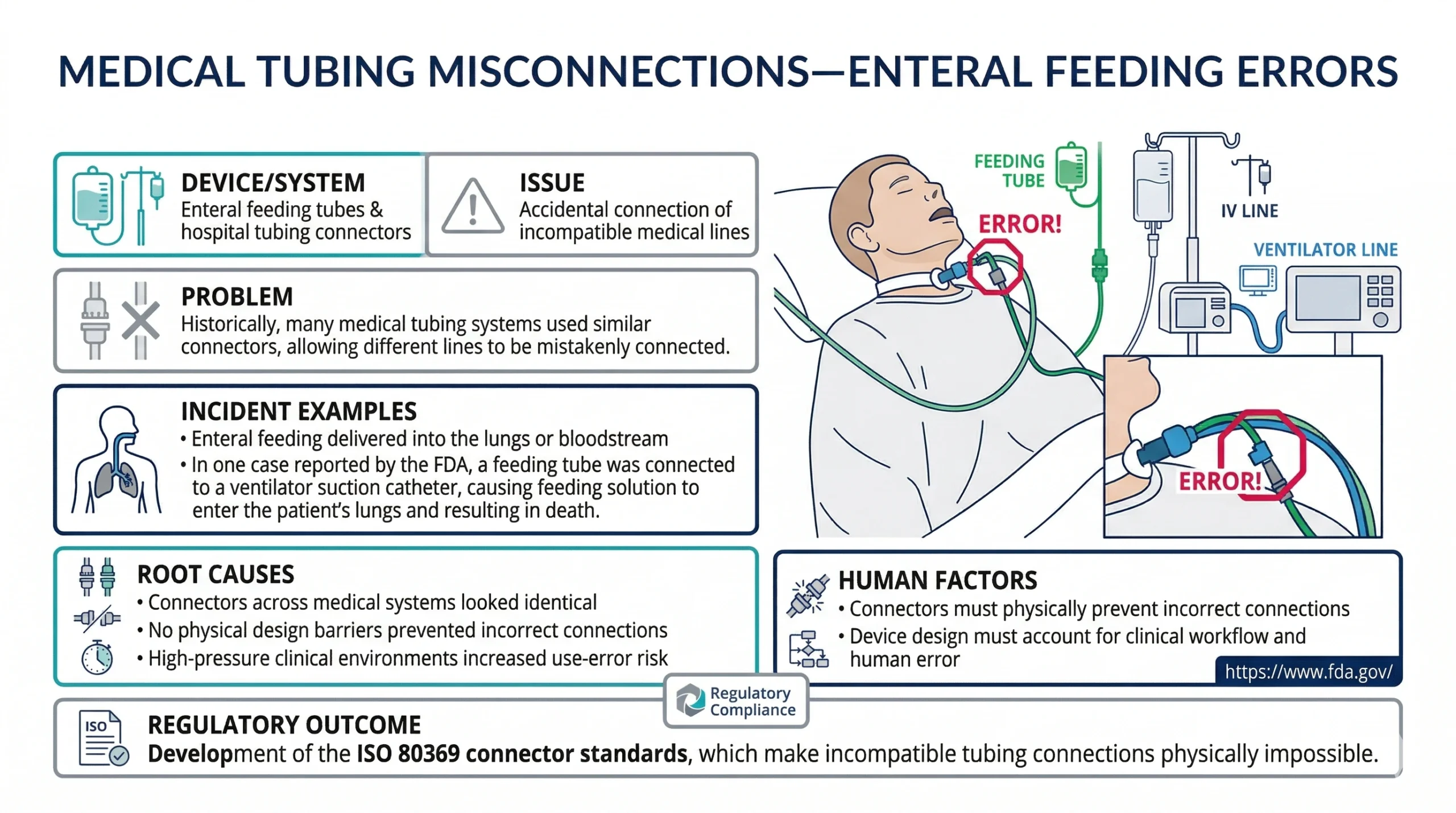
Key Takeaways
Human factors engineering is becoming central to medical device development due to the expansion of home healthcare, increasing software and AI complexity, and growing global regulatory alignment.
Devices used in home settings must accommodate users with varying technical skills and physical abilities, while advanced software introduces new usability challenges related to transparency, trust, and decision support. Regulatory expectations are also converging through frameworks such as FDA human factors guidance, IEC 62366-1, and ISO 14971. Integrating human factors engineering early in development helps ensure safe device use, supports regulatory readiness, and reduces the risk of post-market safety issues by prioritizing user-centerd design.
Insights from the MedTech Guru
Experienced regulatory professionals frequently encounter common questions regarding usability engineering.
Q1: Can training compensate for poor design?
A: Regulators increasingly expect manufacturers to mitigate risks through design improvements rather than relying primarily on user training.
Q2. How many participants are required in a human factors validation study?
A: The FDA does not prescribe a fixed number for all devices, but many validation studies include approximately 15 participants per user group. However, the appropriate sample size depends on several factors, including device complexity, number of distinct user groups, risk associated with incorrect use, and variability in the user population. Manufacturers should justify participant numbers based on study objectives and risk considerations.
Q3. Do software-based medical devices require human factors testing?
A: Yes. In fact, software interfaces are often a major source of usability issues. For software-driven devices, testing focuses on areas such as information presentation, alert interpretation, workflow integration, data entry, and user decision-making, which are closely evaluated during regulatory review.
Q4. How does the shift toward home-use devices affect human factors expectations?
A: Home-use devices introduce unique usability challenges because users often have limited medical training and may face physical limitations. Human factors studies for home-use devices often evaluate scenarios such as:
-
- device setup without professional assistance
- Interpretation of instructions by lay users
- Maintenance tasks such as battery replacement or cleaning
- Environmental factors such as lighting, noise, or distractions
These considerations are increasingly important as healthcare continues to move beyond traditional clinical settings.
Upcoming Events
The MedTech Forum 2026. May 11–13, 2026, Stockholm, Sweden.
MedTech World North America 2026. May 11–13, 2026, West Palm Beach, Florida, USA.
European Medical Device Human Factors and Usability Engineering Conference (EUHF 2026). May 19–21, 2026, Amsterdam, Netherlands.
References
- S. Food and Drug Administration. Applying Human Factors and Usability Engineering to Medical Devices. FDA Guidance for Industry and FDA Staff, 2016.
- S. Food and Drug Administration. Factors to Consider When Making Benefit-Risk Determinations in Medical Device Premarket Approval and De Novo Classifications. FDA Guidance, 2019.
- International Organization for Standardization. ISO 14971: Medical Devices – Application of Risk Management to Medical Devices. ISO, 2019.
- International Electrotechnical Commission. IEC 62366-1: Medical Devices – Application of Usability Engineering to Medical Devices. IEC, 2015.
- Redmill, F., & Rajan, J. Human Factors in Safety-Critical Systems. Butterworth-Heinemann, 1997.
How Can BLA Regulatory Help?
BLA Regulatory, LLC operates as a global regulatory consulting firm specializing in medical device and biopharmaceutical compliance and submission support. With deep experience in U.S. FDA pathways, including IND, NDA, BLA, 510(k), PMA, De Novo, and Breakthrough Device designations, the firm supports sponsors through active agency interactions. BLA Regulatory serves clients across the U.S., Europe, China, and Japan, helping bring safe and effective innovations to market with speed and reliability. For more insights, visit: https://bla-regulatory.com/
Stay in Orbit
Subscribe to future issues of MedTech Orbit for practical updates, U.S. FDA device regulations, and smarter compliance strategies, delivered straight to your inbox.

