2026 User Fees Update! US FDA, EMA, and PMDA
April 16, 2026
Imagine paying 52% more to update your product’s manufacturing label in Europe, or budgeting millions of yen upfront for mandatory meetings before filing a clinical trial notification in Japan. Or facing a year-over-year increase of about $372,001 to file a standard NDA or BLA in the United States. For global sponsors, 2026 brings exactly that reality in comparison to last year.
Planning global regulatory submissions this year will require a keen understanding of shifting financial obligations. Regulatory authorities worldwide are transitioning a larger share of their operational costs to biopharmaceutical sponsors, making accurate financial forecasting essential. Navigating these disparate regulatory economies requires domestic or foreign sponsors to collaboratively sequence their submissions, balancing timeline advantages against cost efficiencies to optimize capital runways and accelerate patient access.
This edition of PharmaFocus covers the 2026 regulatory user fee structures across the US (FDA), Europe (EMA), and Japan (PMDA).
The US FDA: Capacity Planning and Program Stability
The FDA’s user fee programs establish the financial baseline for the American pharmaceutical market. For Fiscal Year (FY) 2026, the agency has published its updated fee structures across all major product categories. These fees reflect baseline inflation adjustments, targeted strategic hiring initiatives, and sophisticated capacity planning methodologies.
Sponsors targeting the US market must budget for high upfront costs. Below is the detailed cost for standard Prescription Drug User Fee Act (PDUFA) fees in FY 2026:
2026 PDUFA Fee Breakdown:
|
PDUFA Fee Category |
FY 2026 Fee (USD) |
|
Application (Requiring Clinical Data) |
$4,682,003 |
|
Application (Not Requiring Clinical Data) |
$2,341,002 |
|
Prescription Drug Program Fee |
$442,213 |
|
Priority Review Voucher (PRV)* |
$1,962,472 |
* In addition to standard marketing applications, sponsors utilizing a Priority Review Voucher (PRV), whether derived from the tropical disease, rare pediatric disease, or material threat medical countermeasure programs, must pay a supplementary utilization fee.
GDUFA, BsUFA, and OMUFA Financial Trends
The financial structures for generics, biosimilars, and over-the-counter (OTC) monographs reflect the unique maturity levels of each respective program.
The Generic Drug User Fee Amendments (GDUFA III) program disperses the financial burden across applications, drug master files (DMFs), manufacturing facilities, and corporate program tiers:
|
GDUFA Fee Category |
FY 2026 Fee (USD) |
|
ANDA |
$358,247 |
|
DMF |
$102,584 |
|
Domestic API Facility |
$43,549 |
|
Foreign API Facility |
$58,549 |
|
Domestic FDF Facility |
$238,943 |
|
Foreign FDF Facility |
$253,943 |
|
Large Size Program Fee |
$1,918,377 |
Conversely, the Biosimilar User Fee Act (BsUFA III) fees for FY 2026 actually decreased across several categories. This downward adjustment reflects the overall maturation of the biosimilar review program and the strategic utilization of available carryover balances:
|
BsUFA Fee Category |
FY 2026 Fee (USD) |
|
Application (Requiring Clinical Data) |
$1,200,794 |
|
Application (Not Requiring Clinical Data) |
$600,397 |
|
Annual Program Fee |
$209,097 |
|
Initial / Annual BPD Fee |
$10,000 |
The modernized Over-the-Counter Monograph Drug User Fee Program (OMUFA II) ensures stable funding for OTC reviews, with adjustments reflecting the complexity of scientific evaluation:
|
OMUFA Fee Category |
FY 2026 Fee (USD) |
|
Monograph Drug Facility (MDF) |
$19,188 |
|
CMO Facility |
$12,792 |
|
Tier 1 OMOR |
$587,529 |
|
Tier 2 OMOR |
$117,505 |
Strategic Considerations for the US
- Zero-Cost Meetings: The FDA does not charge sponsors for standard milestone interactions, such as Type A, B, C, or D meetings. This structure actively encourages early and iterative scientific dialogue without imposing a financial barrier.
- Mandatory Electronic Payments: In accordance with executive mandates to modernize federal banking, the FDA completely phased out paper-based payments as of October 1, 2025. All PDUFA, GDUFA, and BsUFA fees must now be processed electronically via Automated Clearing House (ACH) eCheck, credit card, or wire transfer.
- Foreign Sponsor Risks: Overseas manufacturers must account for international wire transfer delays. If a multi-million-dollar payment does not clear the US Treasury by the submission date, the FDA will issue an immediate Refuse to File (RTF) action, halting the review before it begins. Payments must be initiated at least 5 to 7 business days before your target submission date to ensure full clearance.
European Union (EMA): Implementing the Cost-Based System
In contrast to the FDA’s front-loaded application fee model, the European Medicines Agency (EMA) operates on a strict service-based fee system, charging for nearly every individual regulatory interaction.
The regulatory financial landscape in Europe underwent a systemic change with the enforcement of Regulation (EU) 2024/568, which took effect on January 1, 2025. The regulation replaced fragmented flat-fee models with a workload-based fee system designed to accurately reflect the actual costs incurred by the 29 European Economic Area (EEA) Member States and the EMA itself.
2026 EMA Fee Breakdown:
|
EMA Fee Category |
FY 2026 Fee (EUR) |
|
Type II Variation (Extension of Indication) |
€163,200 |
|
Type II Variation (Standard) |
€22,000 |
|
Annual Fee (Standard Applications) |
€232,400 |
|
Annual Fee (Generic/Hybrid/Informed Consent) |
€60,300 |
|
Annual Fee (Similar Biologic) |
€18,100 |
|
Annual Pharmacovigilance (Per Chargeable Unit) |
€230 |
|
EMA Scientific Advice – Level 1* |
€51,900 |
|
EMA Scientific Advice – Level 2* |
€73,900 |
|
EMA Scientific Advice – Level 3* |
€98,400 |
*These tiers are determined by the breadth and depth of the requested assessment, such as whether the advice covers quality, non-clinical, clinical development, or novel methodologies. Note that the EMA removed the financial distinction between an initial request and a follow-up request. Hence, the standard base fee applies in both scenarios.

Strategic Considerations for the EU
- Payment-Before-Review Policy: Administrative procedures for fee calculations are strictly enforced at validation. Scientific evaluations are explicitly withheld by the EMA until the associated invoice has been paid in its entirety.
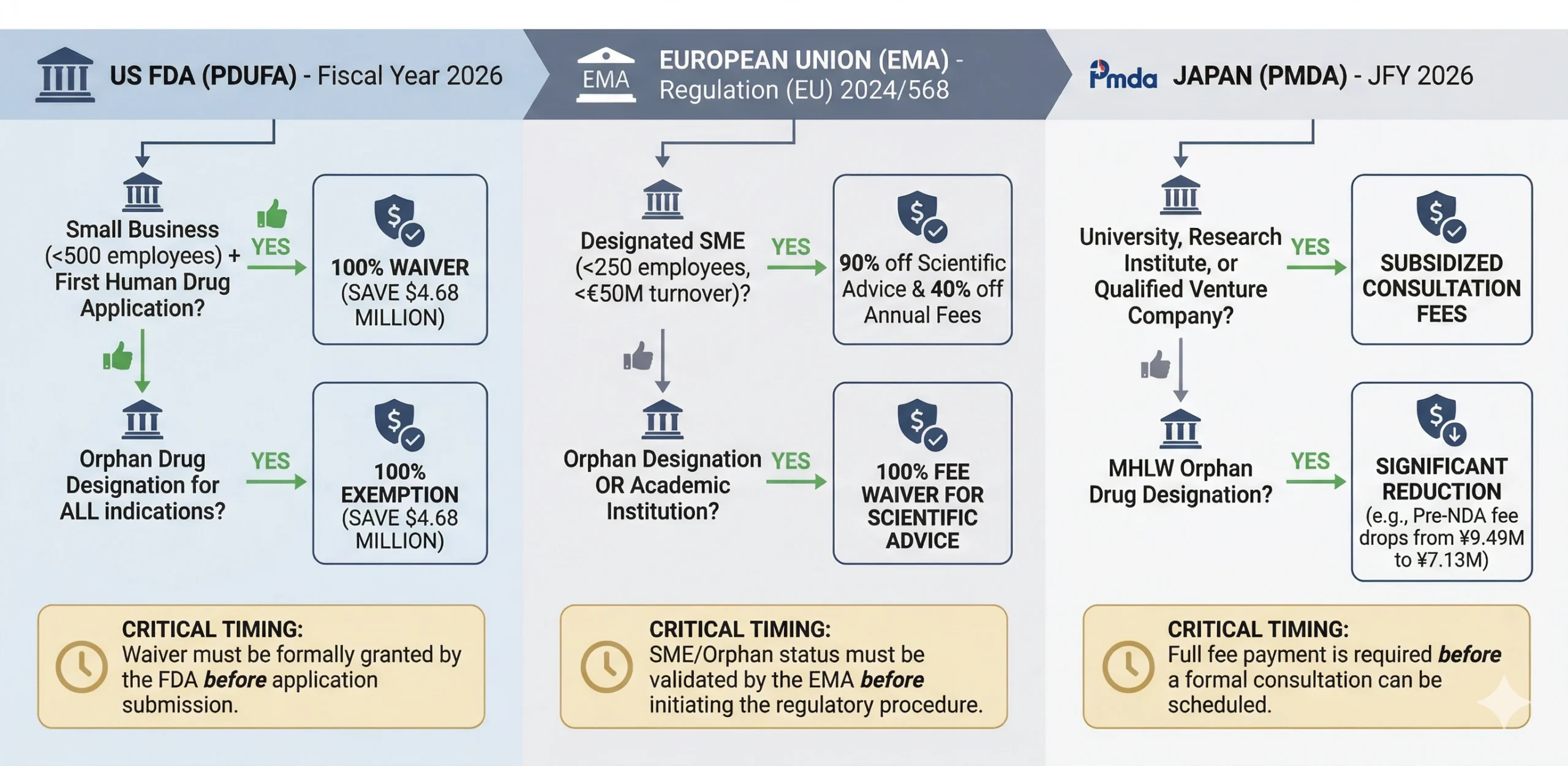
- Fee Reduction Timing: Always secure SME (see last section) or Orphan designations before initiating a Scientific Advice request. Fees are calculated at the exact point of validation. If the status is not confirmed or has expired, sponsors are liable for the full commercial fee.
- Submission Timing Penalties: The EMA applies a pre-submission fee upon the filing of a letter of intent for a marketing application. Subsequent repetitive fees apply if the actual submission date deviates from the intended date by more than 60 days. No fee is charged if a request is withdrawn within 24 hours of submission. However, withdrawals occurring after 24 hours but before the completion of administrative validation will incur a formal administrative charge.
- Minor Variations: Type I minor variations are no longer billed as individual regulatory interactions. These procedures have been consolidated and are now absorbed into the annual fee structure.
Japan (PMDA): Front-Loaded Consultations
Japan’s Pharmaceuticals and Medical Devices Agency (PMDA) offers a highly predictable fee structure that heavily emphasizes paid, upfront consultations rather than just backend application fees.
While marketing application fees exist, the PMDA assesses significant user fees through a structured series of mandatory and highly recommended pre-submission consultations, unlike the FDA, which extracts the vast majority of its user fee revenue at the point of the final marketing application.
For the Japanese Fiscal Year (JFY) 2026, understanding this sequenced consultation model is essential for foreign sponsors engaging the world’s third-largest pharmaceutical market.
2026 PMDA Fee Breakdown:
|
PMDA Strategy Consultation Category |
JFY 2026 Fee (JPY) |
|
RS Strategy Consultation (Drugs) |
¥1,541,600 |
|
RS Strategy Consultation (Medical Devices / Regenerative) |
¥874,000 |
|
Before Start of Phase I (Non-Orphan) |
¥4,578,500 |
|
Before Start of Phase I (Orphan) |
¥3,441,000 |
|
Before Start of Early Phase II (Non-Orphan) |
¥1,752,800 |
|
Before Start of Early Phase II (Orphan) |
¥1,320,200 |
|
After Completion of Phase II (Non-Orphan) |
¥9,497,400 |
|
After Completion of Phase II (Orphan) |
¥7,134,300 |
|
Pre-Application Consultation (Non-Orphan) |
¥9,497,400 |
|
Pre-Application Consultation (Orphan) |
¥7,130,100 |
Strategic Considerations for Japan
- Mandatory Consultation Sequence: The PMDA strictly requires specific, paid consultations before the formal submission of a Clinical Trial Notification (CTN) or a Japanese New Drug Application (J-NDA). This structured sequence ensures regulatory alignment before high-stakes filings.
- Scientific Advice Barriers: The PMDA categorizes its meetings into free pre-consultations and fee-bearing formal Consultations. You use the free pre-consultations to sort out discussion points and confirm your category. If you want formal, actionable guidance on your clinical trial protocols, safety margins, or evidence expectations, you must pay for a formal consultation.
- Escalating Fee Maturity: The financial schedule for Japanese consultations is highly sensitive to the phase of development. As programs progress toward marketing applications, user fees escalate significantly to reflect the increased volume and complexity of the data under assessment.
Global Waiver Strategies: Bypassing the Full Financial Burden
While base regulatory fees are escalating globally, the FDA, EMA, and PMDA all offer critical financial relief pathways. Securing these waivers requires strict adherence to agency-specific criteria and precise timing, often requiring formal validation before a submission is even initiated.
📚 From the PharmaFocus Archives:
- Unlike the FDA, where you pay revenue at the final marketing application, the PMDA assesses heavy user fees through mandatory pre-submission consultations. See our detailed guide on navigating the PMDA consultation timeline: Japan Clinical Trials Unlocked: PMDA Pre-Consultation to First Patient In.
- Securing global waivers requires strict adherence to agency criteria. We have previously detailed how orphan drug (ODD) incentives stack up in Japan, providing a roadmap for rare disease sponsors: Japan’s Legacy in Orphan Drug Development.
How BLA Regulatory Can Help
Miscalculating user fees, missing narrow payment windows, or failing to secure applicable SME or orphan reductions can severely derail submission timelines and drain critical funding, especially when internal teams are not fully aligned across regions.
BLA Regulatory, LLC provides hands-on strategic and operational support across the United States and Japan, with demonstrated capability managing FDA, and PMDA engagements end to end. Our team works directly with sponsors to build comprehensive, multi‑region regulatory budgets, forecast application, facility, program, and consultation fees, and align submission timing with jurisdiction-specific fiscal calendars. We provide on-the-ground expertise in the US and Japan, including direct management of FDA electronic payments and PMDA consultation sequencing and invoicing, to prevent costly Refuse-to-File (RTF) actions or validation delays. We also specialize in identifying eligible fee waivers, securing critical EMA designations, and structuring scientific advice and consultation strategies to maximize regulatory engagement value while protecting capital runway.
References
- Federal Register: Prescription Drug User Fee Rates for Fiscal Year 2026
- FDA: Resource Capacity Planning and Modernized Time Reporting
- EMA: Regulation (EU) 2024/568 Working Arrangements
- EMA: Fees payable to the European Medicines Agency (Human Medicines)
- PMDA: Consultations and Fees for Reviews
- MHLW: Drug Pricing Reform and Pediatric Clinical Trial Frameworks
- Clinical Trial Costs Benchmark (bioaccess)
- FDA User Fees: Examining Changes in Medical Product Development and Economic Benefits (NCBI)