Latest News: Proposed US-FDA Scrutiny on Cross-Border Clinical Data!
May 7, 2026
U.S. policymakers are intensifying scrutiny of foreign involvement in the U.S. drug development and approval process, with a particular focus on clinical trials that support FDA regulatory decision‑making.
On March 19, 2026, U.S. Senator Rick Scott issued a formal letter to the Department of Health and Human Services (HHS), the National Institutes of Health (NIH), and the Food and Drug Administration (FDA), urging immediate action to integrate national‑security considerations into clinical trial oversight.
Importantly, the proposal does not target all clinical data generated outside the United States, but centers on clinical trials conducted under IND or submitted in support of NDA or BLA applications, particularly where such studies involve cross‑border manufacturing, data transfer, or foreign adversary‑linked entities.
Strategic Context: Expanding Biosecurity Mandates
The intersection of clinical research and U.S. national security has escalated rapidly. Since 2024, congressional inquiries have increasingly examined relationships between U.S. biopharmaceutical sponsors and foreign entities linked to strategic competitors, especially in high‑value sectors such as cell and gene therapy.
This is also driven by the economic footprint of the U.S. bioscience industry, noting that in 2023, the sector employed nearly 2.3 million Americans and generated over $3.2 trillion in economic output, indirectly supporting nearly 8 million additional jobs. Protecting this strategic domain has become a paramount priority for U.S. lawmakers.
Initial enforcement actions taken on June 18, 2025, focused on physical and biological components, as explained in our previous analysis of the regulatory environment, PharmaFocus: US FDA Tightens Oversight of Cross-Border Cell Exports!
Legislative attention has since expanded toward digital infrastructure, including clinical trial databases, genomic data repositories, and cross‑border data mirroring practices.
This shift reflects concern not only about data confidentiality but also about the FDA’s ability to exercise effective regulatory oversight, particularly when manufacturing, data processing, or trial operations occur outside the U.S. jurisdiction.
The Catalyst: The Bioheng/Imviva Case Study
Situation
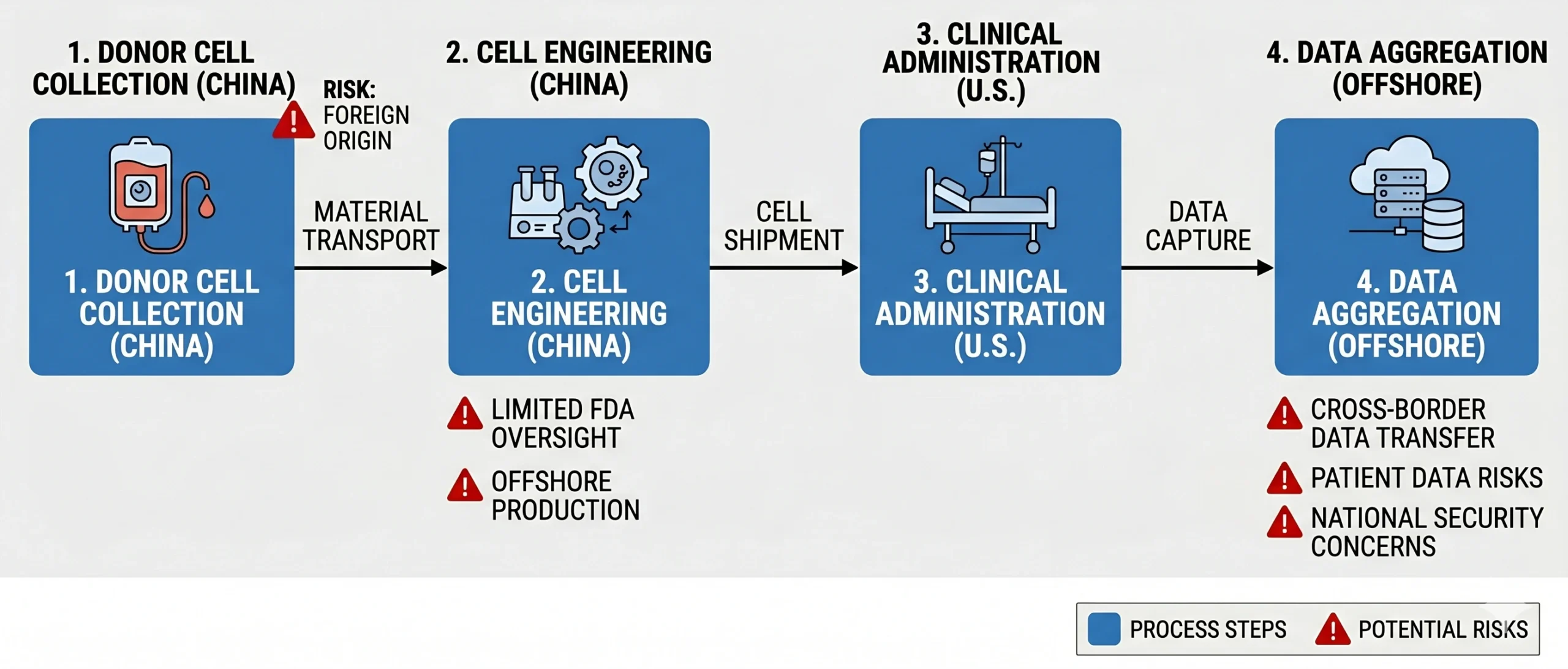
The immediate catalyst for the March 2026 legislative mandate is the regulatory progression of Bioheng, which operates as Imviva Biotech. Imviva is developing CTD402, an allogeneic anti-CD7 CAR-T cell therapy for relapsed/refractory T-cell acute lymphoblastic leukemia (T-ALL) and lymphoblastic lymphoma (LBL).
The program has progressed under a U.S. IND and received Regenerative Medicine Advanced Therapy (RMAT) and Rare Pediatric Disease designations in Oct 2025. In Dec 2025, the first U.S. patient was dosed in the global TENACITY‑01 clinical trial (NCT07070219), followed by FDA Orphan Drug Designation in Jan 2026.
Because this program contributes directly to U.S. regulatory decision‑making, it falls within the category of clinical studies supporting FDA submissions.
Complication
According to publicly available information cited in the Senator’s letter, donor cell collection and CAR‑T manufacturing occur in China, while treatment is administered to U.S. patients and clinical data are transmitted offshore for analysis. Lawmakers have characterized this end‑to‑end cross‑border model as a potential vulnerability, raising concerns around:
- FDA’s ability to conduct effective manufacturing oversight
- Chain‑of‑identity and chain‑of‑custody controls
- Transmission of sensitive U.S. patient data to foreign jurisdictions
Crucially, these concerns arise because the data are used in U.S. regulatory pathways, not solely because of geographic origin.
Ongoing Developments
Highlighting the ongoing intersection of cross-border data flow and scientific advancement, Imviva announced on April 29, 2026, its selection to present updated clinical outcomes for its parallel dual-targeted allogeneic CAR-T candidate, CTA313 (targeting systemic lupus erythematosus), at the American Society of Gene & Cell Therapy (ASGCT) Annual Meeting in Boston, scheduled for May 11–15, 2026.
The case has become a reference point for broader discussions on how global development models intersect with evolving U.S. biosecurity expectations.
What the Senator’s Letter Actually Proposes
The March 19, 2026, letter does not impose a new law, nor does it establish an immediate ban. Instead, it requests administrative action and policy development across seven areas:
1. Immediate Review and Potential Pauses
Lawmakers are demanding an immediate review of all pending and recently approved clinical trial applications involving entities with financial backing, ownership, or significant operational ties to foreign adversaries. Crucially, the request asks agencies to pause approvals where appropriate, pending a comprehensive national security review.
2. Re-evaluation of Expedited Designations
The mandate specifically targets the revocation or reevaluation of expedited designations granted to entities with foreign adversary ties. This includes reassessing the RMAT and Priority Review (PR) status previously granted to Bioheng/Imviva. Sponsors relying on foreign data or manufacturing to secure fast-track, breakthrough, or RMAT designations may need to prepare for intense retroactive scrutiny.
3. Mandatory National Security Screening Protocols
Perhaps the most impactful proposal is the establishment of mandatory national security screening protocols for IND applications and clinical trial proposals. These screenings would be triggered if a clinical trial involves:
- Foreign manufacturing or processing of the therapeutic product.
- The collection, storage, or transmission of patient data outside the United States.
- Sponsorship by entities based in, or controlled by, countries designated as foreign adversaries.
- The use of technologies or methodologies potentially derived from misappropriated intellectual property.
4. Comprehensive Disclosure Requirements
Transparency is heavily emphasized. The proposed framework requires comprehensive disclosure and independent verification of all foreign funding sources and partnerships. Any clinical trial conducted on U.S. soil or involving U.S. patients must also disclose all data-sharing agreements and manufacturing arrangements.
5. Restrictions on Patient Data Transmission
To safeguard sensitive health information, lawmakers seek to restrict the transmission of identifiable U.S. patient data to entities based in or controlled by countries designated as foreign adversaries. Such transmissions would only be permitted under explicit statutory authorization and with robust safeguards in place.
6. Inter-Agency Enforcement Coordination
The FDA, NIH, and HHS are being directed to coordinate actively with the Department of Justice (DOJ), the Federal Bureau of Investigation (FBI), and the broader intelligence community. The goal is to identify researchers, institutions, and companies with undisclosed ties to foreign adversary‑controlled entities and to take appropriate enforcement action.
7. Comprehensive Audit of Active Trials
Finally, the letter demands a comprehensive audit of all active clinical trials involving Chinese entities or researchers with potential CCP ties. This audit aims to assess both national security vulnerabilities and patient safety risks.
The directive imposes a strict 60-day response clock, requiring the agencies to deliver an implementation blueprint by May 18, 2026.
“The United States must remain open to legitimate scientific collaboration, but openness cannot come at the expense of national security or patient protection.”—S. Senator Rick Scott, March 2026
Broader Implications for Global Sponsors
If adopted, these proposals could materially affect sponsors that rely on international manufacturing, global CROs, or cross‑border data architectures, especially in advanced therapeutic areas.
· Heightened Scrutiny on Cell and Gene Therapies
Advanced therapeutics, due to their personalized nature and reliance on genetic data, are at the epicenter of this regulatory shift. The complexities of tracking a patient’s biological material across international borders, maintaining the chain of identity, may be heavily scrutinized. Sponsors must ensure that if processing occurs outside the U.S. jurisdiction, their quality systems can withstand unprecedented FDA auditing aimed at preventing data compromise and guaranteeing manufacturing quality control.
· Increased Disclosure Burden on NIH-Funded Researchers
National Institutes of Health (NIH)-funded researchers have already faced prosecution for failing to disclose foreign affiliations. While federal agencies have recently taken steps to strengthen transparency requirements, lawmakers argue these measures are insufficient to guarantee against national security risks. Sponsors partnering with academic institutions must rigorously vet the financial and collaborative ties of their principal investigators.
· Data Architecture and Localization
The proposed restriction on transmitting identifiable U.S. patient data abroad means that global pharmaceutical companies may need to rethink their data architecture. Implementing robust data localization protocols, where U.S. clinical data remains siloed on domestic servers, may transition from a best practice to a strict regulatory requirement.
· Contractual Qualification Revisions
Standard Master Services Agreements (MSAs) with international contract research organizations and database administrators must be updated. Sponsors must insert clause requirements forcing vendors to completely isolate data infrastructure pathways and provide third-party, double-blind verification of ultimate beneficial ownership matrices.
How BLA Regulatory Can Help
BLA Regulatory supports sponsors in evaluating global clinical development strategies against emerging U.S. biosecurity and regulatory expectations. We build solid IND strategies that form a defensible foundation for future NDA/BLA submissions, conducting targeted risk assessments, mapping cross‑border dependencies, and advising on data localization, vendor qualification, and disclosure preparedness.
References
- Rick Scott Calls for Review of CCP Involvement in U.S. Clinical Trials and Drug Approvals
- Rick Scott Goes To War Over China-Linked Drug Trials In U.S. Clinics – Hoodline
- Senate Special Committee on Aging Hearing: Foreign Dependence on Drug Sourcing
- CLEAR LABELS Act & Congressional Hearings – GovInfo
- Executive Order 14117: Preventing Access to Americans’ Bulk Sensitive Personal Data
- CTD402 Orphan Drug Designation Milestones – CancerNetwork Technical Data
- Bicameral CLEAR LABELS Act Progress and Tracing Provisions – GovInfo Registry
This newsletter is for informational purposes only and does not constitute formal legal or regulatory advice.

