Cracking U.S. FDA Labeling and Claims for MedTech Startups
May 14, 2026
In MedTech, what you say about your device can matter as much as what your device does. The U.S. FDA often determines the intended use of a medical device not only from regulatory submissions, but by looking at the net impression of your labeling and claims, from packaging and instructions for use to website copy, training decks, and sales materials.
For MedTech startups moving fast to win trust, customers, and investors, that creates a real tension: compelling messaging can unintentionally expand intended use, trigger misbranding risk, and pull you into a higher regulatory burden than you planned.
This piece of MedTech Orbit breaks down where companies most often get caught, and a practical way to keep claims ambitious, accurate, and clearance-aligned.
Enforcement Insights- What the Data Shows
Device-specific enforcement by the U.S. FDA, through its Center for Devices and Radiological Health (CDRH), consistently shows that labeling, claims, and intended use are central to regulatory action. In 2025, approximately 30–35 Warning Letters were issued, with a significant portion citing violations related to misbranding, unapproved intended use, or marketing without appropriate clearance.
Review of device Warning Letters (2017–2025) demonstrates top triggers for FDA Warnings:
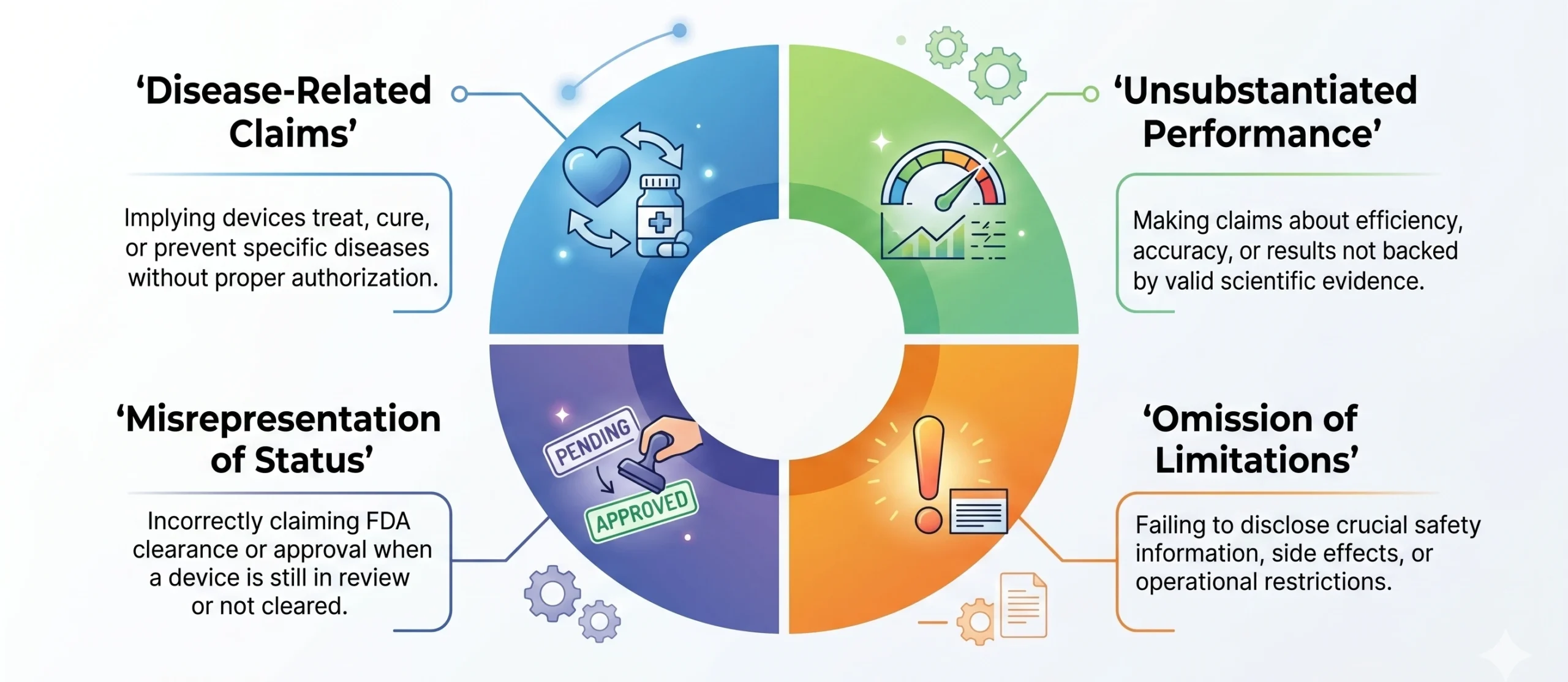
While the FDA does not publish a claims-specific dataset for devices, the consistency of findings across Warning Letters indicates that communication-driven violations are a common enforcement pathway. These actions are typically enforced under statutory provisions for misbranding (Section 502) and marketing without clearance or approval. The evidence underscores a clear regulatory reality: for medical devices, misalignment between claims, evidence, and authorized indications remains a persistent and actively enforced compliance risk.
Labeling and Claims: Regulatory Definitions & Boundaries
Labeling: Function Over Form
Under Section 201(m) of the Federal Food, Drug, and Cosmetic Act and 21 CFR Part 801, labeling includes all written, printed, or graphic material that accompanies a device. The FDA interprets “accompanying” in a functional sense, extending to materials that supplement or explain the product, even if not physically attached.
Accordingly, labeling may include instructions for use, technical documentation, training materials, and digital content. The determining criterion is whether the material contributes to user understanding of the device’s purpose or operation.
Claims: Express Statements and Implied Meaning
Claims encompass both explicit statements and implicit representations regarding a device’s intended use, performance, or clinical benefit. The FDA visualizes claims through a net impression standard, assessing how a reasonable user would interpret the communication as a whole.
This includes direct assertions (e.g., diagnostic capability), comparative positioning (e.g., superiority to standard of care), and contextual cues (e.g., imagery, testimonials, selective data presentation). As a result, compliance requires control not only of language, but of context and presentation.
Regulatory Scope: From Intended Use to Misbranding

Intended Use: Inferred, Not Declared
The FDA determines intended use based on all relevant evidence, including labeling, advertising, and other communications attributable to the manufacturer. This means that a company’s regulatory submission does not exclusively define intended use. External statements may expand or alter it. For example, a disease-related claim, even if isolated, may be sufficient to establish a medical purpose and trigger device classification requirements.
Misbranding: The Standard of “False or Misleading”
Under Section 502(a) of the Act, a device is misbranded if its labeling is false or misleading in any particular manner. This standard encompasses both inaccuracies and omissions.
A claim may be considered misleading if it:
- Lacks adequate scientific support
- Overstates the scope of evidence
- Omits material limitations or risks
- Creates an impression inconsistent with the totality of data
In addition, failure to meet specific regulatory requirements, such as providing adequate directions for use where applicable, may also contribute to a finding of misbranding.

Promotion and Scope of Authorization
Communications that promote a device for uses beyond its cleared or approved indications may be considered evidence of non-compliance. This principle applies regardless of format, including educational content, training sessions, and digital media. While the FDA distinguishes legally between labeling and advertising, both are evaluated in determining intended use and compliance status.
Digital and Third-Party Context
Contemporary enforcement demonstrates that the FDA actively reviews digital environments, including company websites and online platforms. Communications disseminated through distributors, partners, or affiliated channels may also be scrutinized where there is evidence of company influence or control. This reflects an expanded regulatory focus on the ecosystem of communication, rather than isolated materials.
The Claim Compliance Framework
To operationalize compliance, MedTech startups require a structured method for evaluating claims prior to external use. The following framework provides a practical model aligned with FDA expectations.

Key takeaways
In the FDA’s regulatory framework, labeling and claims are not secondary considerations but add to the defining elements of the product itself. Intended use is inferred from communication, and communication determines regulatory obligations.
For MedTech startups, this creates both risk and opportunity. While imprecise claims can trigger enforcement, disciplined and evidence-aligned communication enables credible differentiation in a competitive market.
The strategic imperative is clear: innovation must be matched by precision in how that innovation is described.
Insights from the MedTech Guru
Q: What are the risks of using forward-looking statements?
A: Forward-looking statements can help convey product vision, but if they are not clearly distinguished from current capabilities, they may be interpreted as representing existing functionality when not the case.
Q: How should clinical data be used in communications?
A: Clinical data must be presented with careful context. Without proper framing, it can lead to claims that go beyond what the evidence actually supports.
Q: Do disclaimers protect against misleading claims?
A: Not entirely. While disclaimers can provide useful context, they do not override or neutralize claims that create a misleading or contradictory overall impression. Regulators assess the full message rather than isolated disclaimers.
Q: Is regulatory scrutiny limited to formal submissions and documentation?
A: No. Regulatory authorities also review publicly accessible materials such as websites and digital content, which are common sources of enforcement findings.
Upcoming Events
Digital Health World Congress 2026. May 26-27, 2026, London, UK.
DeviceTalks Boston 2026. May 27-28, 2026, Boston, Massachusetts, USA.
How Can BLA Regulatory Help?
BLA Regulatory, LLC operates as a global regulatory consulting firm specializing in medical device and biopharmaceutical compliance and submission support. With deep experience in U.S. FDA pathways, including IND, NDA, BLA, 510(k), PMA, De Novo, and Breakthrough Device designations, the firm supports sponsors through active agency interactions. Get more insights on device labelling and claims from our team of experts for a smooth submission to regulatory authorities. BLA Regulatory serves clients across the U.S., Europe, China, and Japan, helping bring safe and effective innovations to market with speed and reliability. For more insights, visit: https://bla-regulatory.com/
Stay in Orbit
Subscribe to future issues of MedTech Orbit for practical updates, U.S. FDA device regulations, and smarter compliance strategies, delivered straight to your inbox.