FDA FY 2026 fees are up! Guide to Medical Device Registration & Listings
February 19, 2026
In FY 2026, U.S. market access for medical devices is being defined by execution on fundamentals like Establishment Registration, Device Listing, user‑fee planning, and timely Small Business Determination, rather than by classification alone. Once the regulatory pathway is determined, registration and listing are to be treated as non-negotiable infrastructure, with roles and timing assigned across the supply chain. This MedTech Orbit edition consolidates what must be completed, by whom, and when, so that filings can be scheduled, evidenced, and renewed without avoidable cost or delay.
Device Classification: The True Beginning, Not the Finish Line
Emerging medtech companies often anchor their regulatory strategy on a single question: “What is my device’s classification and pathway?” Classification is essential because it defines the regulatory controls, risk category, and type of premarket submission required.
For a step‑by‑step refresher on how to determine classification and when to use an FDA 513(g), see our companion article: FDA 513(g) & Device Classification Strategy—How to De‑Risk Your Pathway.
Once a company determines its regulatory pathway, two core FDA obligations must be planned for the following classification, regardless of device class or premarket status:
- Establishment registration (who you are & what you do) and
- Device listing (what your product is).
These are required for entities involved in U.S. commercial distribution and allow the FDA to identify who makes/imports devices and which devices are in U.S. commerce
In July 2025, the FDA issued a warning letter to O3UV, LLC for failing to fulfil its annual establishment registration and device listing requirements for Fiscal Year 2025 as required under section 510 of the Federal Food, Drug, and Cosmetic Act. The agency confirmed that O3UV LLC manufactures autohemotherapy and EBOO devices, which are ‘devices’ under the Act, and found that the firm had not submitted its required establishment registration and device listing information for those products. As a result, the FDA determined that the devices were misbranded under section 502(o) of the Act because they were manufactured in an establishment that was not duly registered and were not included in the FDA’s required device listing.
—Warning Letter - O3UV - July 07, 2025
Establishment Registration: Who You Are & What You Do
- Establishment registration is a core legal requirement for medical device entities before commercial distribution in the U.S.
- Under 21 CFR Part 807, registration is role-specific, meaning each organization involved in the development, manufacture, or U.S. importation of a medical device must register independently according to the activities it performs.
- Every entity involved in manufacturing, specification development, sterilization, repackaging, or initial importation must register annually.
Timing: Initial registration within 30 days of starting covered operations.
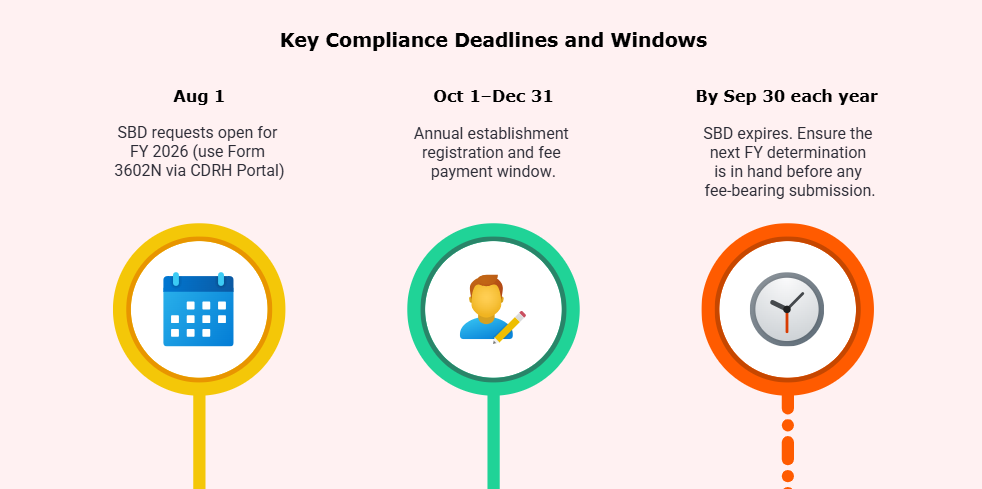
Annual renewal: Between Oct 1 and Dec 31. Device listing kept current with changes

Device Listing — What Is Your Product
- Registration and listing form the operational foundation that allows the FDA to track who makes the device and what devices are available on the U.S. market, independent of classification or marketing authorization.
- Each device placed into U.S. commercial distribution must be listed, including its proprietary name, product code, regulatory status, and the activities performed by each establishment.
- Device listing is required once a device is legally marketed or exempt from premarket review.
FDA User Fees: What Will You Pay (FY 2026)
Effective period: Oct 1, 2025–Sep 30, 2026.
|
Application Type |
Standard Fee |
Small Business Fee† |
|
Annual Establishment Registration Fee |
$11,423 |
|
|
510(k)‡ |
$26,067 |
$6,517 |
|
513(g) |
$7,820 |
$3,910 |
|
PMA, PDP, PMR, BLA* |
$579,272 |
$144,818 |
|
De Novo Classification Request |
$173,782 |
$43,446 |
|
Panel-track Supplement |
$463,418 |
$115,855 |
|
180-Day Supplement |
$86,891 |
$21,723 |
|
Real-Time Supplement |
$40,549 |
$10,137 |
|
BLA Efficacy Supplement* |
$579,272 |
$144,818 |
|
30-Day Notice |
$9,268 |
$4,634 |
|
Annual Fee for Periodic Reporting on a Class III device (PMAs, PDPs, and PMRs) |
$20,275 |
$5,069 |
|
† Small Business Fee: For businesses certified by the Center for Devices and Radiological Health (CDRH) as a small business. For more information, see the section Small Businesses below. |
||
|
‡ 510(k) Fees: All types of 510(k)s (Traditional, Abbreviated, and Special) are subject to the user fee. However, there is no user fee for 510(k)s submitted to the FDA on behalf of an FDA-accredited third-party reviewer. * Certain device BLAs and BLA supplements are administered under MDUFA for CBER-regulated devices and appear in the MDUFA fee table. PMA stands for Premarket Approval (application), PDP stands for Product Development Protocol, PMR stands for Premarket Report, and BLA stands for Biologics License Application. Source: FDA’s MDUFA FY‑2026 fee schedule and Federal Register notice (July 30, 2025) |
||
Small Business Determination (SBD): How to Strategically Save
While registration fees are largely mandatory, submission fees offer significant relief through the SBD Program.
What you may get
- Reduced submission fees if gross receipts/sales ≤ $100M (incl. affiliates).
- One-time “first PMA/PMR/BLA/PDP” submission fee waiver if ≤ $30M (incl. affiliates).
- Registration fee waiver (NEW in FY 2026) if ≤ $1M, prior registration fee paid, and financial hardship (e.g., active bankruptcy). These waivers are discretionary, rare, and evaluated on a case-by-case basis. Startups should plan to pay the full annual registration fee unless explicitly granted relief by the FDA.
Note: FDA currently limits hardship relief to the annual establishment registration fee and does not apply it to initial registration fees.
How & when to apply
- Form FDA 3602N via the CDRH Portal (replaces Forms 3602/3602A) beginning Aug 1, 2025.
- Submit supporting information at least 60 days before the fee is due. Your SBD is valid only until Sept 30 of that FY.
FDA‑registered ≠ Endorsement
FDA and the CFR explicitly warn that registration or possession of a registration number must not be used to imply official approval, as doing so constitutes misbranding (21 CFR 807.39). The FDA also clarifies that “FDA registered/certified” claims and “registration certificates” are misleading and do not mean approved/cleared/authorized. Ensure marketing avoids these claims.
Practical Pitfalls: How to Avoid Them
- Assuming the Contract Manufacturer’s registration covers you: It does not. Each establishment registers independently based on its own role (e.g., specification developer, initial importer).
- Late SBD filings: FDA requires supporting info ≥60 days before the fee is due. If you submit your 510(k)/De Novo/PMA without an approval letter, you’ll pay the standard fee.
- Missing the annual window: Manufacturing or distributing after Dec 31 without completing annual registration leaves you not duly registered, and devices are misbranded.
- Foreign manufacturers without a U.S. Agent: Every foreign establishment must designate a single U.S. Agent. The FDA may treat agent communications as if provided to the firm. Build this into contracting.
Insights from Medtech Guru
Q1. What does the FDA really do when a company misses the December 31 registration deadline?
A1: FDA treats the establishment as not duly registered for the new fiscal year. Any devices manufactured in that establishment and introduced into interstate commerce are legally misbranded under Section 502(o) of the FD&C Act, regardless of prior compliance history or product safety.
Q2. Why is waiting until August to file Form 3602N such a common and expensive mistake?
A2: FDA can take up to 60 days to process Small Business Determination requests, and all approvals expire on September 30. Companies planning an October submission that files in August often miss the cutoff and must submit at the full statutory user fee, even if they otherwise qualify for a reduced rate.
Q3. Why does the FDA view “FDA registered” as a problematic marketing claim?
A3: Establishment registration is administrative, not evaluative. It confirms that the required information was submitted and fees were paid, and does not mean FDA has reviewed, cleared, or approved a device.
Upcoming Events:
- MD&M West 2026. Feb 3–5, 2026. Anaheim Convention Center, Anaheim, California, United States. Regional hub for medical device design, engineering, and manufacturing professionals. https://www.mdmwest.com/en/show-information/show-sectors/medical.html
- Medicall 2026 – Kolkata. Feb 21–23, 2026. Kolkata, West Bengal, India. India’s hospital equipment and medical technology expo with exhibits and industry networking opportunities. https://www.medicall.in/
- MedTech World Middle East 2026. Feb 11–13, 2026. InterContinental Festival City Hotel Dubai, United Arab Emirates. MedTech industry summit with networking and investor/startup sessions. https://med-tech.world/dubai/
- India International Medical Device Expo 2026. Feb 20–23, 2026. HUDA Grounds, Sector 29, Gurugram, Haryana, India. Medical device and healthcare technology expo showcasing products and solutions. https://iitexpo.com/event/medidevice-expo/#
Resource corner:
- MDUFA FY‑2026: Official fees & policy (FDA) [fda.gov]
- Federal Register Notice—FY‑2026 Device User Fees (Jul 30, 2025) [federalregister.gov]
- SBD Program—Reduced or Waived Medical Device User Fees (Form 3602N, CDRH Portal) [fda.gov]
- SBD Final Guidance (Jul 31, 2025)—eligibility, deadlines, hardship waiver criteria [hhs.gov]
- Device Registration & Listing—Who must register/list/pay (FDA) [fda.gov]
- 21 CFR Part 807—definitions, timing, misbranding (incl. §807.39) (eCFR) [ecfr.gov], [ecfr.gov]
- Warning Letter (O3UV, LLC)—registration/listing lapse & 502(o) misbranding [fda.gov]
- FDA explainer—“FDA‑Registered/Certified” ≠ approval (consumer page) [fda.gov]
How Can BLA Regulatory Help?
BLA Regulatory, LLC can provide hands‑on guidance for FDA establishment registration, medical device listing, user fee identification, and Small Business fee qualification—including preparation of required FDA forms, coordination of submissions, and support through the fee‑payment process to ensure accuracy, compliance, and timely completion.
Specializing in medical devices and biopharmaceutical compliance and submissions, the firm brings deep experience across U.S. FDA pathways—IND, NDA, BLA, 510(k), PMA, De Novo, and Breakthrough Device—to support sponsors, startups, and manufacturers throughout the product lifecycle, from strategic planning and gap analysis to meeting materials, agency interactions, and hands‑on submission services. BLA Regulatory serves clients across the U.S., Europe, China, and Japan, helping bring safe, effective innovations to market with speed and reliability.
Stay in Orbit
Subscribe to future issues of MedTech Orbit for practical updates, U.S. FDA device regulations, and smarter compliance strategies— delivered straight to your inbox.

