US FDA CMC Strategy: CGT Comparability and BLA Readiness
March 19, 2026
Comparability has become a defining regulatory risk for cell and gene therapy (CGT) programs as they approach late-stage development. Over the past several years, US FDA review experience increasingly shows that comparability gaps, rather than isolated Chemistry, Manufacturing, and Controls (CMC) deficiencies, are driving regulatory delay and review friction. Manufacturing change is unavoidable in CGT development, driven by the need for process optimization, scale-up, and preparation for commercialization. This edition of PharmaFocus examines how domestic and foreign sponsors can approach CMC comparability as a comprehensive lifecycle strategy, beginning early and extending through Biologics License Application (BLA) readiness, thereby mitigating clinical holds and preserving development timelines.
Bridging US FDA and EMA Expectations
For domestic and foreign sponsors alike, navigating the divide between the US FDA and the European Medicines Agency (EMA) is a primary strategic hurdle. While both authorities ground their expectations in ICH Q5E (Comparability of Biotechnological/Biological Products), the application of these principles to Advanced Therapy Medicinal Products (ATMPs) varies in nuance.
The EMA’s Committee for Advanced Therapies (CAT) often emphasizes a stepwise approach to comparability, as detailed in their Reflection paper on the regulatory requirements for the development of ATMPs (EMA/CAT/852615/2018). While the US FDA and EMA both require a demonstration that changes do not adversely affect product quality, the EMA frequently places a higher premium on the comparability exercise as a formal, stand-alone protocol submitted via Scientific Advice.
Our Advice: Foreign sponsors must realize that while the US FDA may accept a risk-based justification in an Investigational New Drug (IND) amendment, the EMA may require more extensive head-to-head data even for mid-stage process shifts. Alignment between these two bodies is increasing, but sponsors must still design comparability protocols that satisfy the most stringent requirements of both regions to ensure global market readiness.
Regulatory Trends in CGT Comparability
The regulatory landscape for advanced therapies is shifting rapidly. As more CGT programs advance toward pivotal trials and BLA submission, the FDA has signaled limited tolerance for retrospective, minimally justified, or purely analytical comparability approaches.
Recent Complete Response Letters (CRLs) and late-cycle review comments from the FDA indicate heightened scrutiny of comparability strategies, particularly where process evolution affects product potency, cellular heterogeneity, or clinical relevance. The FDA explicitly evaluates manufacturing changes against 21 CFR 601.12, which mandates that a sponsor must demonstrate that a manufacturing change does not adversely affect the identity, strength, quality, purity, or potency of the product. Sponsors can no longer afford to treat comparability as a checkbox exercise executed just before submission and consider strategically weaving it into the clinical development plan from the outset.
“It is our hope that by providing robust guidance in this area, we can help sponsors make plans for how they will move forward from the outset, so that ultimately, we avoid delays at the end of development as people try to catch up and show that the products with which they have completed their pivotal studies are the same as those with which they originally started.”— Dr. Peter Marks, Director, FDA Center for Biologics Evaluation and Research (CBER)
Multi-Attribute Methods (MAM) and Orthogonality
The industry is moving away from a one-test-for-one-attribute mindset toward Multi-Attribute Methods (MAM). MAM, typically utilizing high-resolution Liquid Chromatography-Mass Spectrometry (LC-MS), allows sponsors to monitor multiple Critical Quality Attributes (CQAs), such as post-translational modifications, purity, and sequence integrity, in a single injection.
However, with this increased sensitivity comes increased regulatory scrutiny. The US FDA now expects a strategy of orthogonal testing. This involves using two or more different scientific principles to measure the same attribute. For example, if you are measuring viral vector aggregation, relying solely on Dynamic Light Scattering (DLS) may no longer be sufficient. The FDA may expect orthogonal data from Analytical Ultracentrifugation (AUC) or Size-Exclusion Chromatography with Multi-Angle Light Scattering (SEC-MALS) to verify the results. This double-verification approach ensures that manufacturing changes haven’t introduced subtle artifacts that a single method might miss.
Why Comparability Is Uniquely Challenging in CGT
For traditional, well-characterized recombinant biologics like monoclonal antibodies, sponsors rely heavily on extensive analytical characterization to meet the comparability standard. However, for CGT products, the inherent biological variability of living cells, complex and often manual manufacturing systems, and limited batch history create a challenging environment for proving sameness.
For instance, the inherent starting material variability in autologous cell therapies (patient-to-patient differences) creates a high degree of background noise. This noise makes it incredibly challenging to detect process-related shifts through standard analytics alone. The FDA formally acknowledged these unique challenges in its Draft Guidance on Manufacturing Changes and Comparability for Human Cellular and Gene Therapy Products (July 2023). The agency notes that manufacturing changes in CGT may introduce risks that cannot be fully mitigated through analytical testing alone, often necessitating a weight of evidence approach that includes non-clinical and sometimes clinical data.
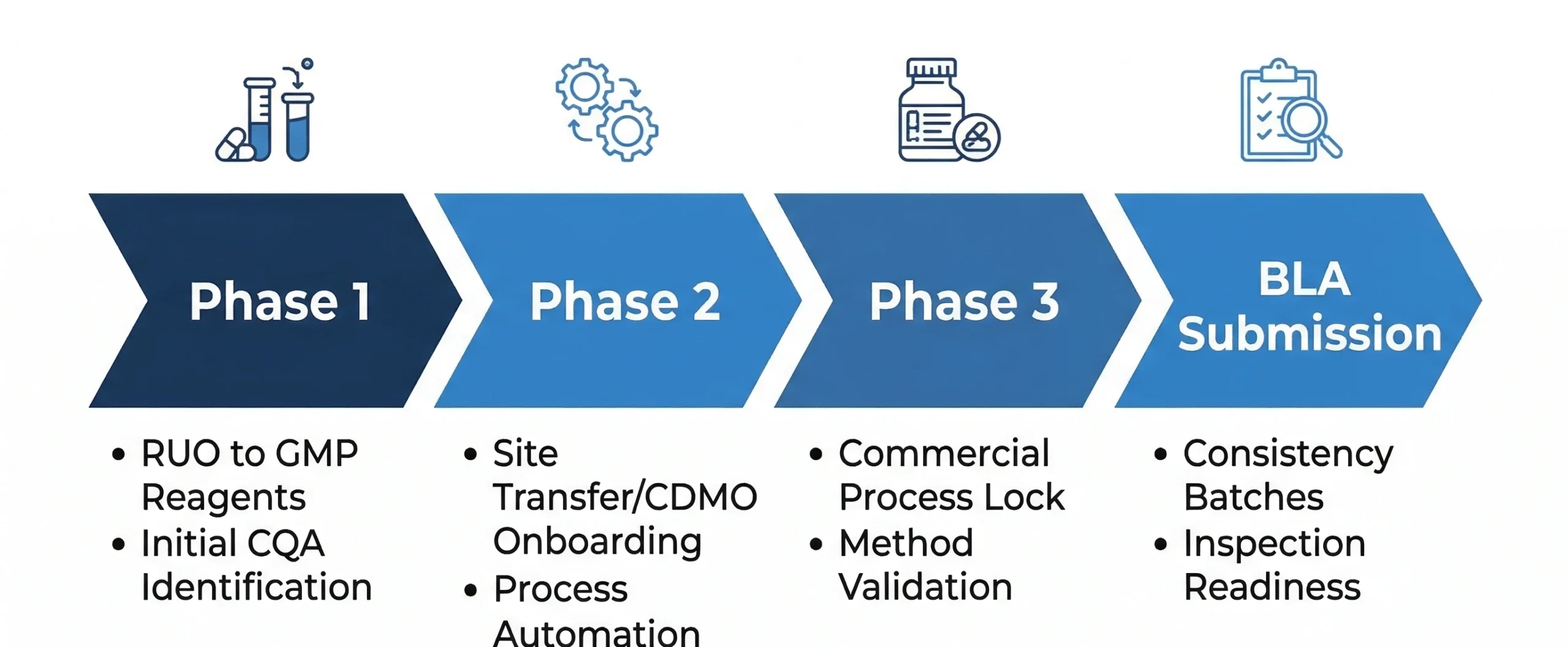
Early Development: Designing for Future Comparability
During Phase 1 and early Phase 2, sponsors implement foundational changes: cell bank replacement, vector platform modifications, and the transition from research-grade to GMP-grade raw materials. Transitioning from Research Use Only (RUO) reagents to GMP-grade materials is a high-risk inflection point. Many sponsors assume this is a simple upgrade, but a change in reagent source can significantly alter the cellular phenotype or impurity profile.
The FDA expects sponsors, even at early stages, to generate sufficient data to understand product quality. The regulatory standard, reinforced by the FDA’s Considerations for the Development of Chimeric Antigen Receptor (CAR) T Cell Products (January 2024), emphasizes that robust early characterization mitigates future risk. Establishing baseline analytical profiles and retaining reference material are foundational requirements for later-stage comparability success.
Mid-Stage Development: Scaling Without Breaking Comparability
As CGT programs enter Phase 2/3, manufacturing changes typically shift from incremental adjustments to structural modifications with higher regulatory impact. Platform changes and scale or site transfers, in particular, represent inflection points where FDA expectations for comparability evidence increase materially.
At this stage, FDA’s focus increasingly shifts from individual changes to the cumulative risk profile of manufacturing evolution. Sponsors are expected to assess manufacturing changes based on their potential impact on critical quality attributes (CQAs) using formal risk assessment methodologies like Failure Mode and Effects Analysis (FMEA).

Scenario 1: The Phase 3 Site Transfer Misstep
• Situation: A sponsor developing an autologous CAR-T cell therapy achieved excellent clinical readouts in Phase 2 and transferred manufacturing to a larger commercial facility for their pivotal Phase 3 trial.
• Complication: The sponsor relied primarily on routine release testing to prove comparability. However, differences in bioreactor fluid dynamics and shear stress at the new facility caused an undetectable shift in the T-cell memory phenotype (specifically, a decrease in stem cell memory T-cells). The FDA placed a clinical hold under 21 CFR 312.42, noting that routine assays did not address the potential impact on in vivo persistence and long-term efficacy.
• Resolution: The sponsor suffered a severe delay of over 10 months. They were forced to develop advanced flow cytometry characterization assays and retroactively test retained Phase 2 material against the new batches to prove clinical equivalence before the trial could resume.
Late-Stage and BLA Readiness: Where Programs Succeed or Fail
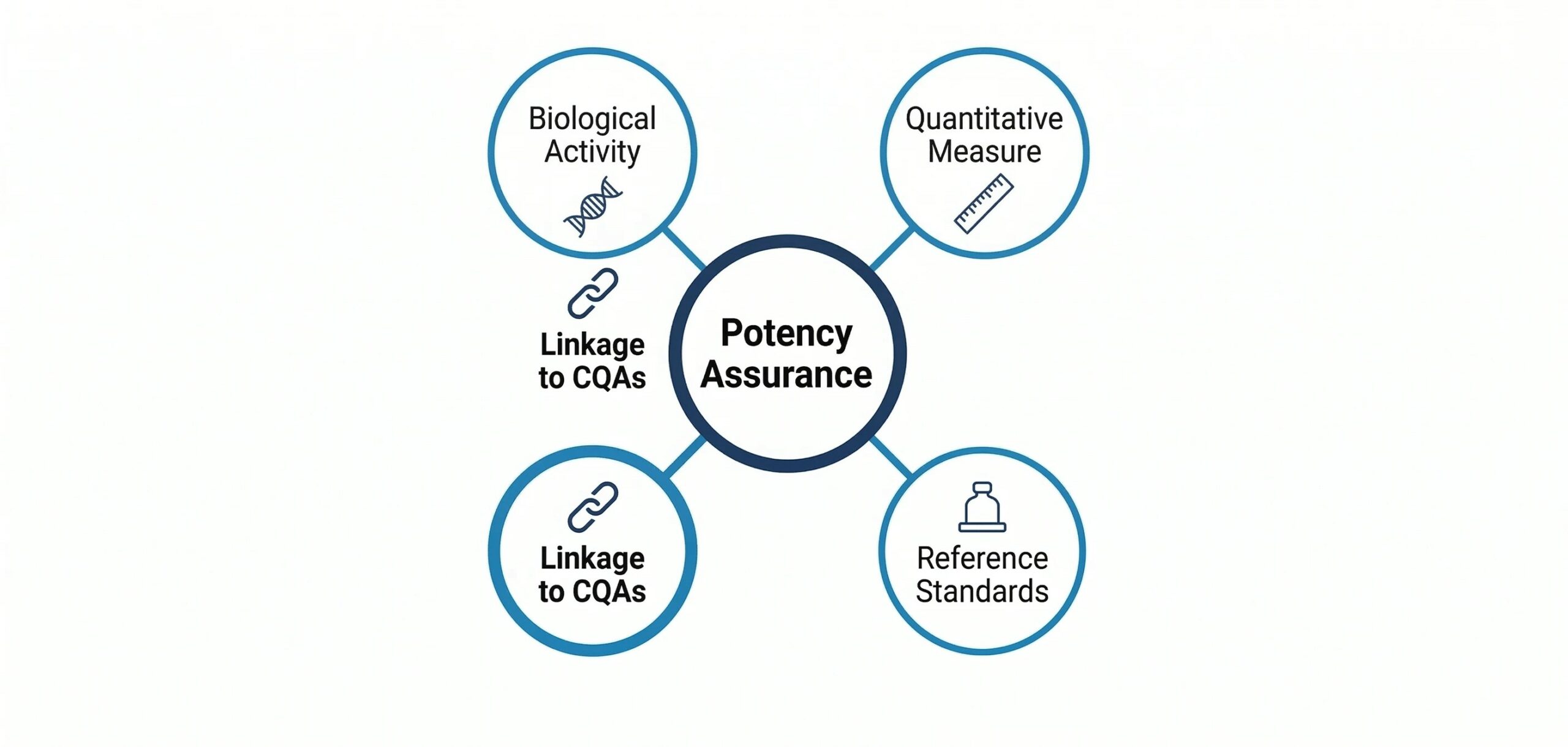
For BLA submissions, under 21 CFR 601.2, an application must provide robust data demonstrating that the manufacturing process reliably produces a safe, pure, and potent product. The FDA recently modernized its approach in the Draft Guidance on Potency Assurance for Cellular and Gene Therapy Products (December 2023).
This guidance shifts the paradigm from simple potency testing to a holistic potency assurance strategy grounded in quality risk management. If a sponsor optimizes a potency assay late in development, they must conduct rigorous bridging studies to ensure the new assay accurately reflects the biological activity established during pivotal trials.
Statistical Rigor in Comparability
A common pitfall in late-stage submissions is the lack of statistical depth. Demonstrating sameness in CGT requires more than just comparing means. As programs mature, the FDA looks for robust statistical approaches, such as Quality Attribute (QA) Acceptance Criteria or, where sample sizes permit, Equivalence Testing. Sponsors must define their comparability acceptance criteria (CAC) before testing begins, ensuring they are based on historical process capability and the clinical relevance of the attribute.

``If there is not a concurrent CMC development going on with that rapid progression of the clinical development, CMC will fall behind... At the end of the day, very late in the stage, there will be a major problem waiting for that development program.``Dr. Steven Oh, Deputy Director, FDA CBER Office of Therapeutic Products (OTP)
Scenario 2: Managing Vector Platform Modifications
• Situation: A gene therapy sponsor transitioned from an adherent cell culture to a scalable suspension-based process for adeno-associated virus (AAV) vector production between Phase 1 and Phase 2.
• Complication: While viral titer increased significantly, the ratio of full-to-empty capsids shifted. The sponsor initially failed to correlate how this change in capsid distribution might impact the product's immunogenicity profile and in vivo potency.
• Resolution: Through an INTERACT meeting, the sponsor proactively developed an orthogonal assay strategy using Analytical Ultracentrifugation (AUC) to precisely quantify capsids and implemented tighter purification steps. The FDA accepted this scientific justification and the revised control strategy, allowing the program to proceed without the need for extensive animal bridging studies.
Navigating Complex Comparability Hurdles: Q&A
Q: Does analytical comparability failure always require a new clinical trial?
A: No. A risk-based evaluation determines if non-clinical in vitro or in vivo bridging studies can fill the gap. Clinical trials are typically a last resort reserved for cases where fundamental mechanism-of-action or safety profiles have changed.
Q: Can we use literature data to justify a comparability gap if our samples are gone?
A: Rarely. The FDA expects product-specific data. Depleted reference samples are a major CMC oversight that literature data cannot fully rectify, often leading to requests for new bridging data.
Q: How does the FDA view the transition from RUO to GMP reagents?
A: The FDA expects this transition to be managed through a formal risk assessment. If the reagent is a critical component (e.g., a cytokine in cell therapy), you must prove that the GMP version does not alter the product’s quality or cellular characteristics.
BLA Regulatory partners with CGT sponsors to eliminate regulatory uncertainty by designing forward-looking comparability strategies that align with both US FDA and EMA expectations. From implementing Multi-Attribute Methods (MAM) and orthogonal testing strategies to preparing for critical INTERACT and Type B/C agency meetings, we actively audit late-stage comparability data and potency assurance strategies. Our goal is to resolve critical gaps before they trigger a Complete Response Letter, positioning your product for a rapid, seamless path to BLA approval.
References
- FDA. Draft Guidance: Manufacturing Changes and Comparability for Human CGT Products. July 2023.
- FDA. Draft Guidance: Potency Assurance for CGT Products. December 2023.
- EMA. Reflection paper on regulatory requirements for ATMPs. 2018.
- FDA. Considerations for the Development of CAR T Cell Products. January 2024.
- ICH Q5E. Comparability of Biotechnological/Biological Products. June 2005.
- 21 CFR 601.12 – Changes to an approved application.
- 21 CFR 312.42 – Clinical holds and requests for modification.
- 21 CFR 601.2 – Applications for biologics licenses.

