Ready for US-FDA E2B(R3) Safety Reporting from April 1st?
March 12, 2026
Effective April 1, 2026, the U.S. Food and Drug Administration (FDA) will make the use of the ICH E2B(R3) electronic format mandatory for all Individual Case Safety Report (ICSR) submissions and will no longer accept legacy format filings after this date.
Implementation began in early 2024, covering postmarketing reports in January and premarketing data in April. With a mandatory compliance deadline of April 01, 2026, sponsors are expected to align their pharmacovigilance (PV) systems. The intention of transitioning from legacy E2B(R2) format to this granular XML-based structure is to enable unified safety surveillance and enhanced signal detection. Our experts think this is an upgrade in how the industry is capturing and transmitting critical patient safety data. In this edition of PharmaFocus, we break down what you need to know.
Understanding the Basics

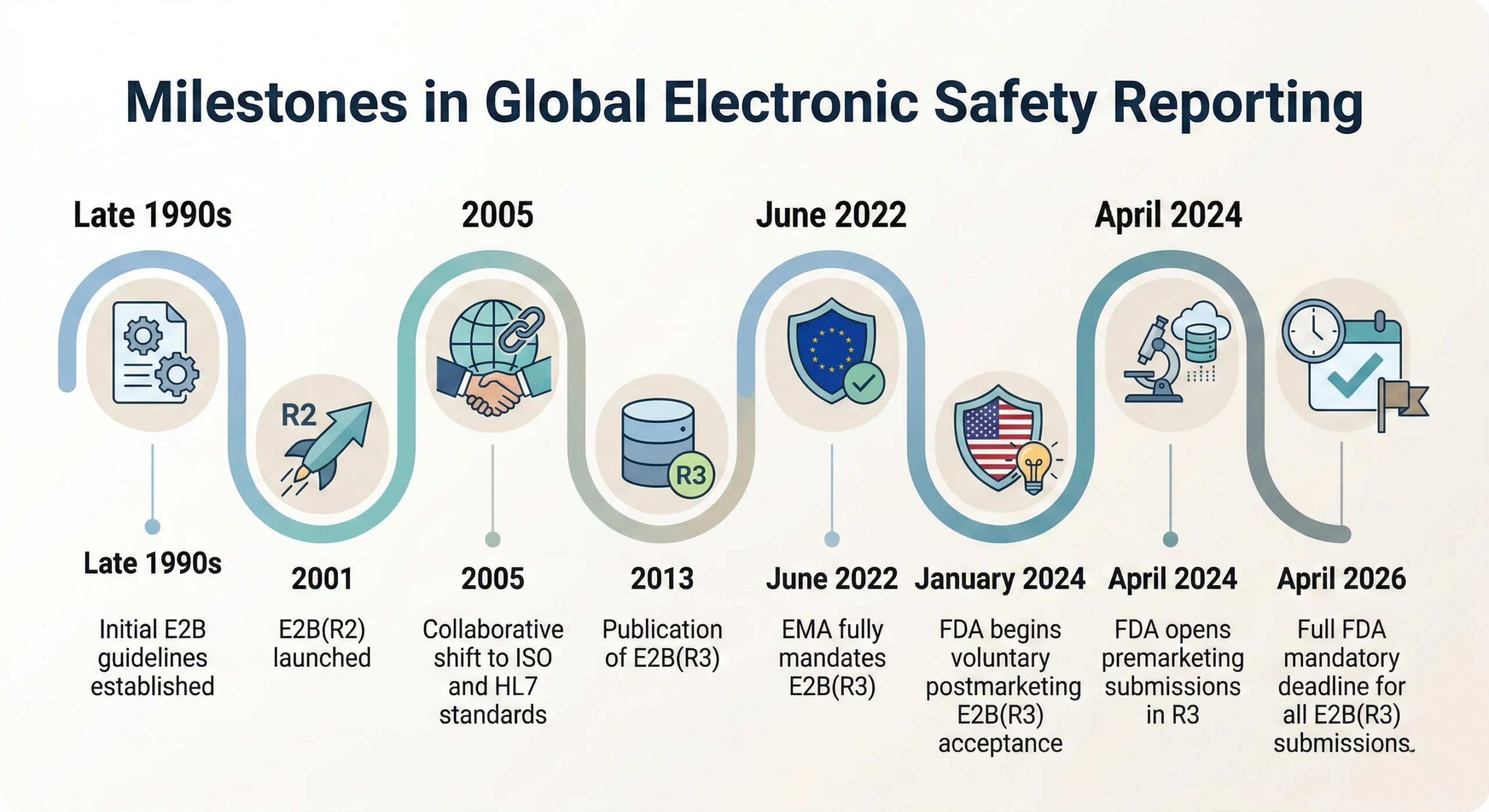
Milestones in Global Electronic Safety Reporting
The shift you see below shows that the U.S. FDA remains aligned with international health authorities, including the European Medicines Agency (EMA) and Japan’s Ministry of Health, Labor and Welfare (MHLW), who have also confirmed or implemented the adoption of E2B(R3).
Premarketing Impact: The IND
Under 21 CFR 312.32(c)(1)(i), sponsors of clinical trials conducted under an IND are required to report serious and unexpected suspected adverse reactions (SUSARs) in an IND safety report to the FDA and participating investigators. Historically, these were submitted via the Electronic Common Technical Document (eCTD) as PDF files. However, as of April 1, 2024, the FDA has opened the door for these reports to be submitted in the E2B(R3) format directly to the FDA Adverse Event Reporting System (FAERS).
This change is outlined in the final guidance for industry, Providing Regulatory Submissions in Electronic Format: IND Safety Reports, published in April 2024. Sponsors had 24 months from the publication of this final guidance to comply with electronic submission requirements, setting the mandatory deadline at April 1, 2026. Here is a quick reference snapshot:
|
Submission Type |
Regulatory Requirement |
Electronic Submission Format |
|
IND Safety Reports |
E2B(R3) XML |
|
|
BA/BE Safety Reports |
Voluntary E2B(R3) or PDF |
|
|
Periodic Safety Reports |
eCTD via ESG |
|
|
Postmarketing ICSRs |
E2B(R3) XML |
Critical Nuance: Not all IND safety reports qualify for the E2B(R3) XML structure. Reports detailing findings from other studies, animal or in vitro testing results, or observations of increased incidence rates still require eCTD submission. These reports require contextual narrative explanation and study interpretation that cannot be fully captured within standardized XML fields.
Our recommendation: Sponsors must therefore maintain two distinct submission pathways: the E2B(R3) gateway for patient-level ICSRs and the eCTD pathway for aggregate or non-clinical safety findings, such as your annual periodic safety reports (DSUR, IND Annual Report).
Postmarketing Reporting: NDA, BLA, ANDA
For drug products marketed under approved New Drug Applications (NDAs), Abbreviated New Drug Applications (ANDAs), or Biologics License Applications (BLAs), the electronic submission of postmarketing safety reports has been mandatory since the final rule issued on June 10, 2014. The implementation of E2B(R3) for postmarketing reports began on January 16, 2024. During the transition period ending April 1, 2026, the FDA did permit the continued use of the E2B(R2) standard for postmarketing ICSRs to allow sponsors to upgrade their safety databases. The goal of this modernization is to house all IND studies and post-marketing data in one source, which will greatly enhance the efficiency of signal detection and quality reviews across product classes.
Critical Nuance: Once a sponsor chooses to submit their first E2B(R3) report for a specific product, they are technically locked into that standard. After April 1st this year, this applies to premarketing individual reporting as well. We have received FDA confirmation that a company may not revert to legacy methods or standards once the transition has begun.
Our recommendation: This no-reversion policy makes careful planning and exhaustive validation a necessity before the first production file is transmitted, considering the short turnaround timelines for individual safety reporting.
What does this mean for Global Sponsors and PV Teams
- Regulatory Alignment and Strategic Implications: For sponsors operating internationally, this transition reduces duplication of effort and supports a unified approach to global regulatory compliance.
- System and Infrastructure Readiness: Organizations may need to collaborate with software vendors or internal IT teams to implement necessary upgrades, validate data mappings, and ensure seamless integration with existing workflows.
- Operational Pathways for Submission: Under the new mandate, sponsors have two primary options for submitting ICSRs in E2B(R3) format:
-
- Automated electronic transmission via the FDA’s ESG, or
- Manual entry through the Safety Reporting Portal (SRP).
The ESG pathway is ideal for organizations with high reporting volumes and established IT capabilities, offering efficiency and scalability. Conversely, the SRP provides a more accessible alternative for smaller entities or those with limited technical resources, though it still requires familiarity with the E2B(R3) data structure and adherence to the FDA’s technical conformance guidelines.
- Enhanced Responsibilities for PV Teams: PV teams must be trained to navigate these expanded requirements, identify and correct data inconsistencies, and ensure that all mandatory fields are populated to avoid submission rejections. This may also involve revising internal SOPs and enhancing collaboration between clinical, regulatory, and IT functions.
- Risk Mitigation and Compliance Planning: Establishing cross-functional task forces and investing in staff training will be essential to building organizational competence and confidence in the new reporting paradigm. Proactive engagement with the FDA and industry working groups can also provide valuable insights and best practices during the implementation phase.
Navigating Challenges during Transition
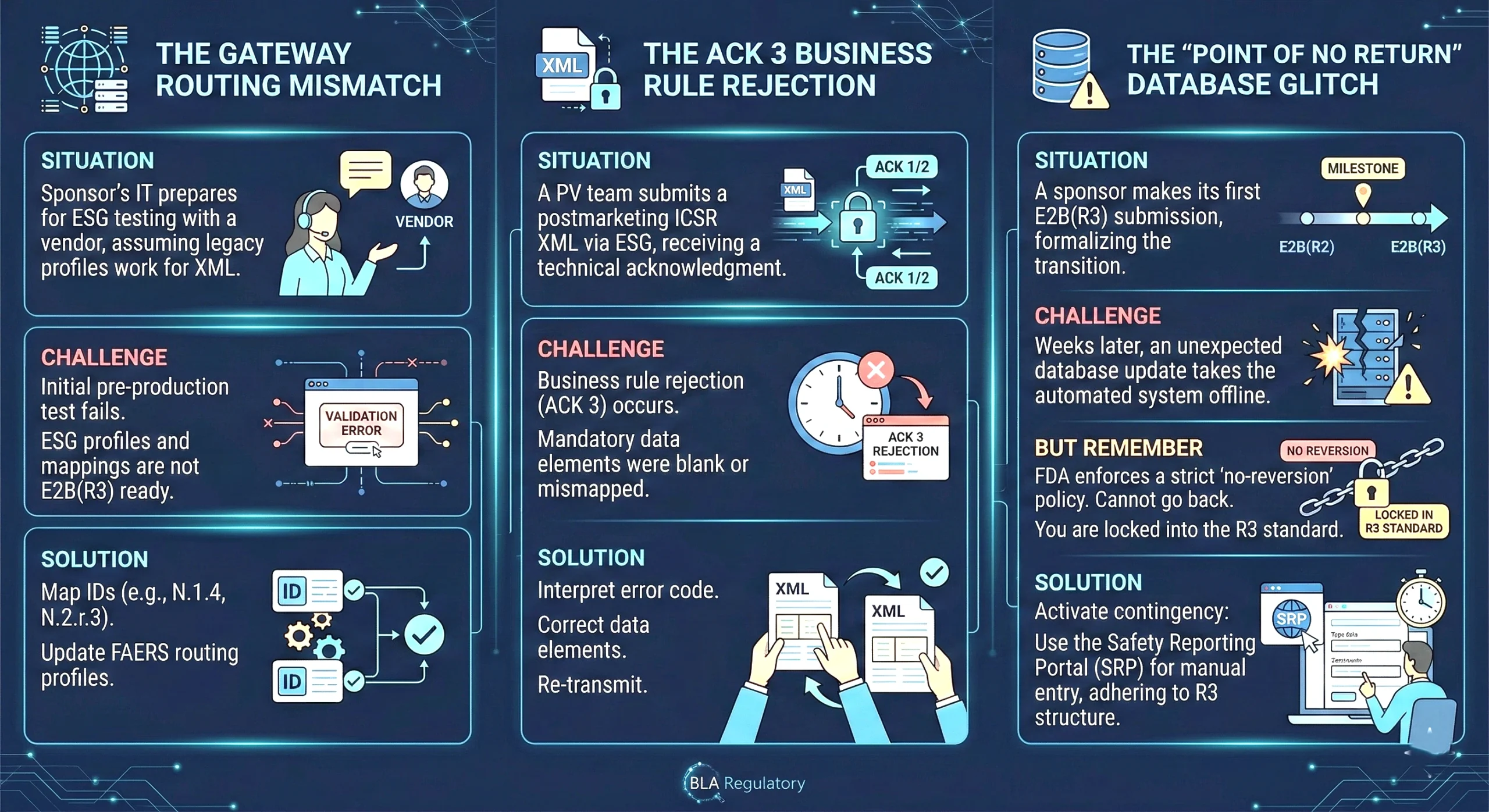
What Needs to Be Done Before April 1, 2026
- System Audit: Confirm that your safety database is configured with the latest FDA E2B(R3) profiles.
- Account Registration: Ensure active accounts for both the FDA Electronic Submissions Gateway (ESG) and the WebTrader platform if not already established.
- Gateway Routing: Map accurate Batch Receiver Identifiers (N.1.4) and Message Receiver Identifiers (N.2.r.3) for both pre- and post-market report types
- Pre-Production Testing: Conduct end-to-end testing in the pre-production environment to ensure your XML files pass both schema and FDA-specific business rule validations.
- Policy Update: Revise Internal SOPs to account for the no-reversion policy once E2B(R3) submissions begin.
Note: If a third‑party safety database vendor or external regulatory technology provider manages your ESG/WebTrader setup or routing configurations, reach out and confirm that they are fully prepared for post‑production E2B(R3) submissions, including correct routing IDs, up‑to‑date FAERS profiles, and readiness to meet FDA business rule validations.
Our Take on the End-to-End E2B(R3) Submission Process
If we had to manage the workflow of this new requirement, this is how it would look:
- Case Intake: Collection of safety information from spontaneous reports, clinical trials, literature, partners, or support programs.
- Case Processing: Medical review and coding using MedDRA and WHO Drug, followed by seriousness assessment, causality evaluation, and data quality checks.
- E2B(R3) Message Generation: Mapping all case data to the required E2B(R3) elements and generating the standardized HL7 XML file according to FDA’s technical specifications.
- Regulatory Transmission: Secure electronic submission through the appropriate pathway (ESG or SRP), ensuring adherence to the FDA’s regional implementation guidelines. Monitoring and interpreting technical (ACK 1/2) and business rule (ACK 3) responses, resolving any validation errors or rejections promptly.
- Ongoing Case Maintenance: Submitting follow-ups, corrections, or nullifications as additional information becomes available or discrepancies are identified.
How Can BLA Regulatory Help?
BLA Regulatory streamlines your transition to E2B(R3) by managing both test and production submissions on your behalf, ensuring validation accuracy, submitting through ESG or SRP, and tracking acknowledgments end-to-end. This lets sponsors meet the 2026 mandate without investing in complex internal infrastructure. As we continue to grow, BLA Regulatory is planning to expand its capabilities to include in-house E2B(R3) file generation and enhanced pharmacovigilance support, enabling sponsors to benefit from a more comprehensive, integrated safety reporting solution.
References
- ICH E2B(R3) Clinical Safety Data Management: Data Elements for Transmission of Individual Case Safety Reports
- FDA Regional Implementation Specifications for ICH E2B(R3) Implementation: Post market Submission of Individual Case Safety Reports for Drugs and Biologics, Excluding Vaccines
- Providing Submissions in Electronic Format—Postmarketing Safety Reports
- Microsoft Word – E2B R3 Reporting Scenarios
- Reporting premarket and postmarket safety reports to FDA using ICH E2B(R3) standards
- Preparing for the electronic exchange of safety reports
- E2B(R3) Electronic Transmission of Individual Case Safety Reports (ICSRs) Implementation Guide – Data Elements and Message Specification
- FDA E2B(R3) Core and Regional Data Elements and Business Rules
- ICH and FDA regional data elements E2B(R3) Reporting Scenarios
- E2B(R3) Individual Case Safety Report (ICSR) Specification and Related Files

