Welcome to the First Edition of MedTech Orbit – An Expert’s Guide for Medical Device Sponsors & Manufacturers to Navigate U.S. MedTech Regulations!
Whether you’re preparing to launch your first medical device in the U.S. or expanding an established portfolio, understanding FDA requirements is essential to your success. This newsletter is here to deliver clear, timely insights that help sponsors and manufacturers bring safe, effective, and compliant devices to market — smarter and faster.
In this inaugural issue, we start with the basics: navigating the U.S. FDA regulatory landscape.
Medical devices are governed by diverse regulatory systems worldwide. Europe’s Medical Device Regulation (MDR) has elevated requirements around clinical evidence, post-market surveillance, and cybersecurity. Health Canada follows its own licensing framework; Japan, China, and others tailor their regulations to local health needs and infrastructure.
Even amid this global patchwork, the U.S. FDA stands out as a benchmark. Approval by the FDA often signals credibility in international markets—and many countries reference U.S. regulatory decisions when shaping their policies. So, whether you’re a global player or focused solely on the U.S., understanding the FDA’s approach sets you up for success.
Global & G20 Outlook: Why the U.S. Market Matters
https://market.us/report/medical-devices-market/
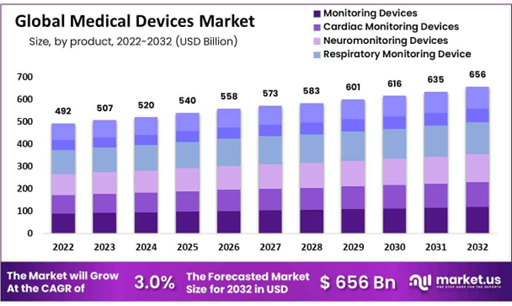
The medical devices market in G20 countries is projected to reach around $438.3 billion in 2025. Of that, the U.S. alone accounts for $200.14 billion—the largest single market within G20. The domestic medical device manufacturing sector alone is estimated to support over 530,000 jobs. Further, the U.S. medical device market is forecasted to grow to over $312 billion by 2027, representing a 5.4% CAGR.
These figures are more than just numbers—they signal the wealth of regulatory opportunity in the U.S. market. No wonder companies worldwide prioritize FDA compliance early in their strategy.
The FDA’s Medical Device Classification Ladder
The FDA groups medical devices into three risk-based classes. Think of it like a ladder—the higher you climb, the more risk is involved, and the more scrutiny the FDA applies.
Class
|
Risk Level
|
Examples (Catheters)
|
Regulatory Requirements
|
Why It Matters
|
| Class I — Simple & Safe |
Low |
Vas-cath, pericardiocentesis catheter, set & tray.
Product code: JCY |
Generally, exempt from premarket submissions.
Must comply with general controls (labeling, facility registration, GMP, complaint reporting). |
Device registration and listing take around 1–2 weeks.
Low-risk but still FDA-regulated.
Errors in quality or labeling can cause recalls and harm the reputation. |
| Class II — Similar but Stronger |
Moderate |
Coronary infusion catheter, continuous flush (e.g., K953133) |
Require 510(k) premarket submission demonstrating substantial equivalence.
Must comply with general controls. |
Clearance takes 3–6 months.
Strategic data preparation is key to avoiding delays. |
| Class III — Serious Business |
High (life-sustaining, critical devices) |
Catheters, transluminal coronary angioplasty, percutaneous (e.g., P860019) |
Require Premarket Approval (PMA) — FDA’s most rigorous review.
Extensive clinical data are required to prove safety and effectiveness. |
Clearance takes 2–5 years.
PMA route is resource- and time-intensive; critical for high-risk devices used in implantable or advanced applications. |
Classification Beyond Labels
Device classification impacts everything—from the type of evidence needed (technical documents for Class I, testing for Class II, clinical studies for Class III) to time-to-market (weeks vs. months vs. years) and costs (thousands for 510(k) vs. millions for PMA). Getting the classification right early is critical, as misclassification can cause delays, wasted resources, and block market entry. The FDA’s 513(g) program offers a formal way to request early feedback—giving clarity before committing time and resources to the wrong regulatory route.
Device approval trends
Each year, thousands of Class I medical devices are registered and listed with the FDA. However, the majority of these are exempt from formal premarket review and do not require clearance. In contrast, Class II and Class III devices must typically undergo more rigorous FDA review processes, including 510(k) or PMA submissions.
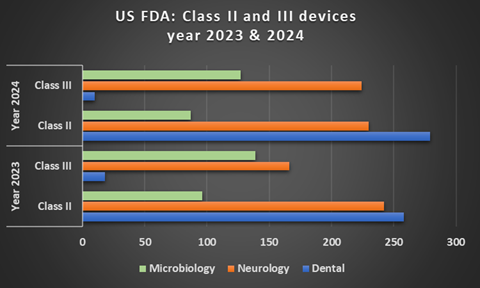
The FDA does not publish a complete annual count of Class I device listings or exemptions, but data on Class II and III device approvals can be accessed through public FDA databases. Approval volume also varies significantly by device panel. For example, in 2023 and 2024, the cardiovascular panel accounted for a high number of clearances and approvals, while panels such as microbiology, neurology, and dental had comparatively fewer—reflected in the chart below.
Information derived from 510k database and PMA database, FDA
A few misconceptions often trip up new entrants in the medical device space. One of the most common is the belief that all devices require clinical trials. In reality, only Class III devices—and occasionally certain novel Class II devices—typically need extensive clinical data. Many products move forward on the strength of bench testing and predicate comparisons alone. Another frequent misunderstanding is that Class I devices are “unregulated.” While they may be exempt from premarket review, they are still subject to the FDA’s general controls, including labeling requirements, facility registration, and good manufacturing practices. Finally, some assume that FDA approval is the end of the journey. The truth is- regulatory responsibility doesn’t stop at clearance or approval. Companies must maintain robust quality systems, remain inspection-ready, and actively monitor device performance throughout the product’s lifecycle.
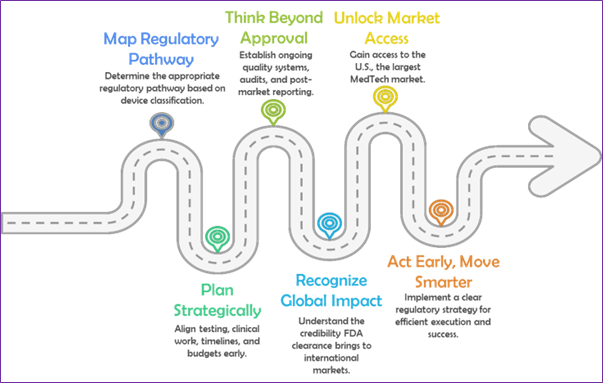
With your device’s classification and regulatory path clearly defined, the next step is execution: aligning teams, timelines, and technical work with FDA expectations. That means being deliberate—investing in the right testing, anticipating documentation needs, and planning for compliance from day one.
But regulatory success doesn’t stop at market entry. Ongoing FDA requirements—like quality systems, reporting, and audits—demand sustained attention. Treating compliance as a continuous process, not a one-time hurdle, protects both your product and your company’s reputation.
Ultimately, FDA clearance or approval does more than unlock access to the U.S. market. It sends a signal of credibility to investors, partners, and regulators worldwide—accelerating your path to global scale.
Insights from the MedTech Guru
Stay connected and informed with these upcoming regulatory and MedTech opportunities:
Conferences & Summits
- https://indiamedexpo.com/ime/
India Med Expo, Hyderabad, India (5-7 Dec 2025)
- Global Conference on Pharma Industry and Medical Devices
Global Conference on Pharma Industry and Medical Devices, Bangkok, Thailand (12-13 Dec 2025).
- https://www.mdassn.org/MD2025/
6th International Conference on Medical Design,Taoyuan, Taiwan (19-22 Dec 2025)
- https://ifearp.org/Conference/13737/WCPIMD/
World Conference on Pharma Industry and Medical Devices, Abu Dhabi, UAE (13-14 Jan 2026)
- https://www.arena-international.com/event/octmedeurope/
14th Annual Outsourcing in Clinical Trials: Medical Devices Europe, Munich, Germany (27-28 Jan 2026)
- MedTech Dubai Summit – MedTech World
MedTech Event (11-13th Feb, 2026)
BLA Regulatory, LLC., operates as a global regulatory consulting firm specializing in medical devices and biopharmaceutical compliance and submission support. With deep experience in U.S. FDA pathways—including IND, NDA, BLA, 510(k), PMA, De Novo, and Breakthrough Device designations—the firm supports sponsors, startups, and manufacturers through the full product lifecycle: from strategic regulatory planning and gap analysis to preparation of meeting materials, agency interaction, and hands-on submission services. BLA Regulatory serves clients across the U.S., Europe,China, and Japan , and —helping bring safe and effective innovations to market with speed and reliability. For more insights visit: https://bla-regulatory.com/
Subscribe to future issues of MedTech Orbit for practical updates, U.S. FDA device regulations, and smarter compliance strategies— delivered straight to your inbox.