U.S. FDA Tightens Oversight of Cross-border Cell Exports!
April 23, 2026
On June 18, 2025, the U.S. FDA suspended certain new clinical trials involving the transfer of American citizens’ living cells to six designated “countries of concern” for ex vivo genetic engineering and subsequent re-administration in the United States. Framed as a biosecurity and patient safety measure, the action represents a significant tightening of regulatory scrutiny over cross‑border cell processing and reverses prior exemptions that allowed offshore sample handling under defined conditions.
This edition of PharmaFocus examines the regulatory basis for the FDA’s enforcement approach, assesses its implications for global clinical trial design, and outlines practical compliance strategies for U.S. and foreign sponsors navigating this evolving policy landscape.
Background and Evolution of the FDA Ban
The policy shift should be understood as part of a broader, multi-year tightening of U.S. biosecurity controls (see timeline below).
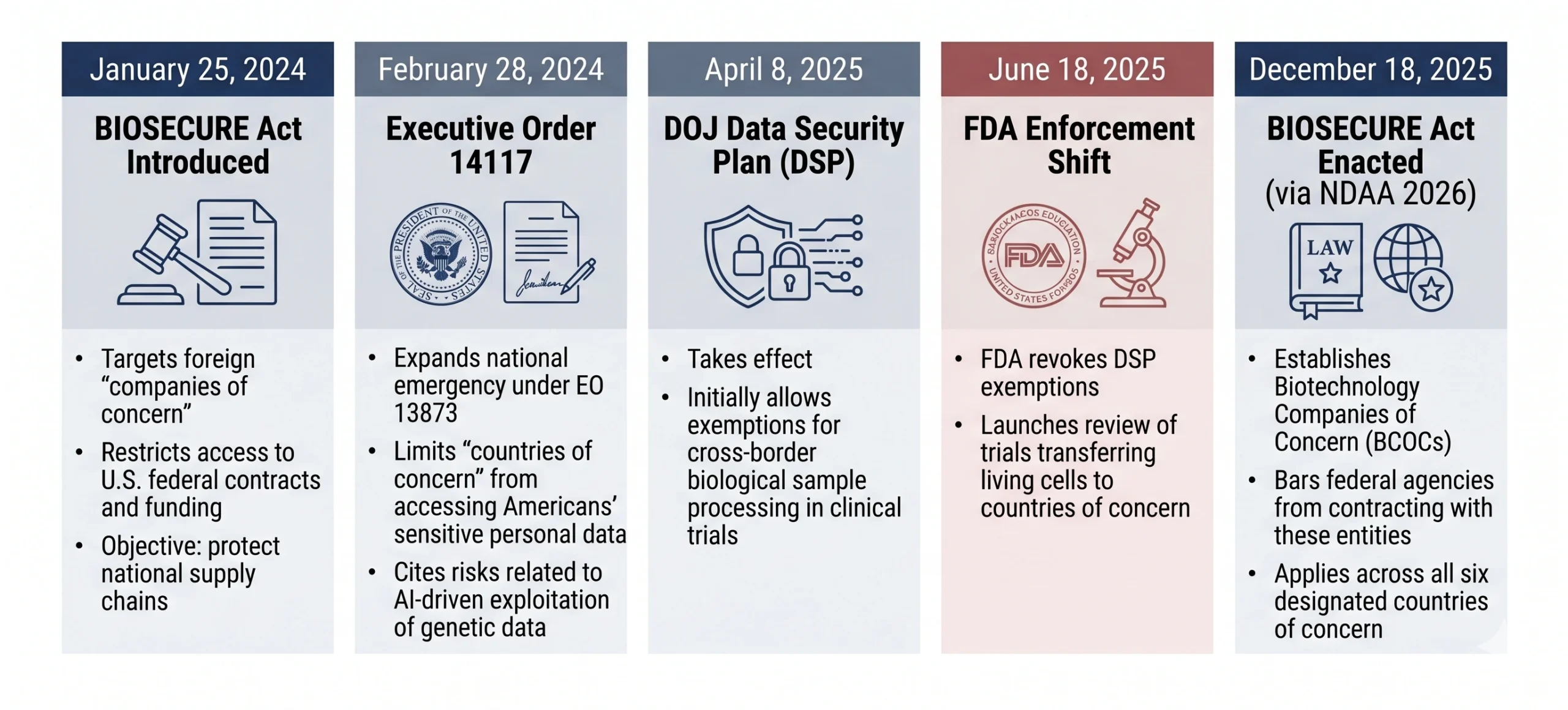
Mechanism of Enforcement: The Clinical Hold!
The FDA is issuing clinical holds on new trials and, in some cases, evaluating ongoing trials where the supply chain involves exporting patient cells (e.g., leukapheresis products) to designated countries of concern for ex vivo genetic engineering.
Under 21 CFR 312.42(b)(1)(i), the FDA may place an IND on clinical hold if “human subjects are or would be exposed to an unreasonable and significant risk of illness or injury.”
The Agency is interpreting unreasonable and significant risk to encompass the unauthorized access, exploitation, or misuse of a patient’s fundamental genetic material by hostile foreign actors.
Types of Cells and Genetic Engineering Technologies Involved
The current status of enforcement heavily impacts the advanced therapies sector. The therapies most vulnerable to these specific clinical holds include:
-
- Chimeric Antigen Receptor (CAR) T-cell Therapies: Autologous therapies requiring the collection of patient T-cells, international shipment for viral vector transduction, and return shipment for infusion.
- CRISPR/Cas9 and Gene Editing: Therapies requiring ex vivo genetic manipulation of hematopoietic stem cells or other patient-derived tissues.
- Personalized Cancer Vaccines: Modalities relying on the sequencing of a patient’s tumor and normal tissue to engineer bespoke immunotherapies in foreign facilities.
Definition and Scope of “Countries of Concern”

These countries are identified as:
- engaging in a persistent pattern of conduct or serious incidents harmful to U.S. national security and the safety of U.S. persons.
- posing a significant risk of using government-related data or bulk sensitive personal data to harm U.S. national security or the safety of U.S. citizens.
Notably, this definition stands in contrast to other national security regulatory regimes. For example, Executive Order 14105 on foreign investment only names China. The national security guardrails of the U.S. Department of Commerce’s CHIPS program only name China, Russia, Iran, and North Korea, and the Department of Commerce’s rules on information and communications technology and service supply chains only name China and Russia.
For Foreign Sponsors: Compliance Challenges and Response Strategies
Core Responsibilities and New Requirements for Sponsors
Under 21 CFR Part 312, sponsors bear ultimate responsibility for trial quality, integrity, and ethics. The FDA’s new policy adds three core obligations for collaborations involving “countries of concern”:
- Enhanced Transparency in Informed Consent: Explicitly disclose cell transfer destinations, processing methods, and genetic data exploitation risks, including region-specific threats.
- Sample and Data Localization: Process sensitive biological materials in the U.S. or non-restricted regions, terminate partnerships with labs in restricted nations, and validate new compliant facilities.
- Proactive Regulatory Engagement: Conduct pre-trial FDA consultations to confirm compliance, including documenting supply chain restructuring away from all restricted nations.
Adjustment Strategies for Clinical Trial Design and Execution
- Mandate Multi-Regional Clinical Trials (MRCTs): Design trials with U.S. patients and exclude sites in restricted nations, e.g., redirecting Cuba-reliant immunotherapy trials to Spanish or Mexican sites.
- Mitigating Reagent Shortages from Russia: Sponsors relying on Russian bioreagents (e.g., specialized cell culture media) are qualifying European alternatives (e.g., German-based Miltenyi Biotec), though this increases costs by 15-20%.
- Strengthening Data Governance: Given varying data protection standards across “countries of concern,” sponsors are implementing unified data management systems that comply with FDA, EMA, and GDPR requirements, eliminating cross-border transfers to restricted nations entirely.
Impact Assessment on Costs and Timelines
The direct economic impact of the FDA ban on sponsors is significant:
- Increased Trial Execution Costs: Using multiple laboratories for sample transportation, data coordination, and other activities may cost sponsors over $1.2 million annually.
- Supply Chain Restructuring Costs: Transferring work originally outsourced to Chinese CDMOs to other regions or within the United States is expected to incur substantial conversion costs.
- Prolonged Trial Cycles: While the FDA’s statutory IND review timeline is 30 days, sponsors note that restructuring supply chains and pre-IND engagement requirements may extend effective start-up timelines relative to China’s implied approval system of 45 days.
The integrity of our biomedical research enterprise is paramount. We are taking action to protect patients, restore public trust, and safeguard US biomedical leadership.– FDA Commissioner Dr. Marty Makary, FDA Press Release, June 2025
For U.S. Pharma Companies: Business Impact and Strategic Adjustments
Current Status and Challenges of Overseas R&D Layout
U.S. pharmaceutical companies have extensive R&D collaborations with China, including major deals between AstraZeneca and Chinese biotechs, and Kite Pharma’s licensing agreement with Shenzhen Prgen Biotech. However, the FDA ban and the BIOSECURE Act are disrupting this landscape. The BIOSECURE Act designates initial BCOCs like WuXi AppTec and BGI and empowers the Office of Management and Budget to add more entities within 365 days.
Supply Chain Risks and Cost Pressures
U.S. biopharmaceutical companies face severe supply chain risks:
- High Reliance on Chinese CDMOs: U.S. biopharmaceutical companies outsource 30% of the workflows for FDA-approved drugs to Chinese CDMOs. Both large enterprises, such as Eli Lilly, and small biotech companies, such as lovance Biotherapeutics, rely on services from WuXi Biologics.
- Supply Risks of APIs and KSMs: China’s dominant position in Active Pharmaceutical Ingredients (APIs) and Key Starting Materials (KSMs) poses significant risks to the U.S. drug supply chain, with nearly a quarter of U.S. generic drugs potentially originating from China.
- Cost Structure Shifts: Phase III trials cost $98,000 per patient in China (vs. $123,000 in the U.S.). Chinese CROs cut costs to 1/3 of U.S. counterparts, and China’s gene therapy products are 60% cheaper than international equivalents.
Impact on Innovation Capacity and Market Competitiveness
The FDA ban has complex impacts on the innovation capacity of U.S. pharmaceutical companies:
- Positive Impacts:
- Promote the reconstruction of U.S. domestic biopharmaceutical manufacturing capacity. Incentives such as tax credits, advanced market commitments, and streamlined FDA processes may begin to address long-standing health security gaps.
- Facilitate cooperation with other countries such as India, South Korea, Israel, and Europe to achieve supply chain diversification.
- Negative Impacts:
- Small cell and gene therapy companies relying on foreign manufacturing will be adversely affected.
- China’s advantages in early-stage drug development may persist. According to PitchBook, a widely cited industry database for biopharma deals, there were at least 6 cancer-related transactions in January 2026 alone, including a deal by AbbVie with a potential value of nearly $6 billion.

Looking Ahead
FDA’s current enforcement posture reflects an active, ongoing review process, rather than a single, time‑bound action.
- Case‑by‑case regulatory engagement:
FDA has not announced a blanket rulemaking or formal ban. Instead, sponsors are expected to address concerns through pre‑IND meetings, IND amendments, and direct engagement to demonstrate compliant manufacturing, data handling, and informed consent practices. - Alignment with existing Executive Orders:
The enforcement approach aligns with broader federal actions under Executive Order 14117 and related national security and data protection measures. These authorities do not set fixed implementation deadlines but enable ongoing interagency oversight of sensitive biological materials and associated data flows. - No formal guidance issued to date:
As of now, FDA has not released new guidance documents or compliance deadlines specific to overseas cell processing. Sponsors should therefore expect continued reliance on existing IND authorities, sponsor responsibilities under 21 CFR Part 312, and evolving FDA feedback during regulatory interactions - Implication for sponsors:
Regulatory expectations are being shaped through enforcement and review rather than formal rulemaking. Sponsors should plan for continued scrutiny, document supply‑chain decisions carefully, and engage FDA early when overseas cell processing or genetic engineering is involved.
📚 From the PharmaFocus Archives:
FDA Expands Surprise Inspections at Overseas Manufacturing Sites (Aug 20, 2025)
Explores intensifying FDA inspections of foreign CDMOs with emphasis on cell handling risks and supply‑chain restructuring. Read more »
How BLA Regulatory Can Help
BLA Regulatory supports sponsors navigating FDA’s increased scrutiny of overseas cell processing by assessing regulatory risk associated with cross‑border cell and data flows, advising on compliant manufacturing and supply‑chain strategies, and supporting FDA engagement through pre‑IND meetings, IND amendments, and Type B or C interactions. Our team assists with drafting and updating IND documentation, protocols, and informed consent language to address FDA expectations on cell handling, data governance, and transparency, while monitoring enforcement trends and policy developments to help sponsors anticipate and manage evolving regulatory risk.
References
- FDA Halts New Clinical Trials That Export Americans’ Cells to Foreign Labs in Hostile Countries for Genetic Engineering | FDA
- S. Government Publishing Office. (2024). Executive Order 14117 of February 28, 2024.
- Uncertainty remains over FDA block on China engineering of US cells
- Overseas cell processing may take a hit as FDA reviews trials
- Politics Over Patients? FDA’s Ban on Overseas Sample Processing Undermines U.S. Biomedicine
- Fierce Pharma Asia—AZ-CSPC deal; FDA’s American cell shipping ban; China’s trial review acceleration plan
- FDA to Review Clinical Trials That Utilize Overseas Cell Processing
- First subject dosed in trial of GenEditBio’s GEB-101 for corneal dystrophy
- ProRelix Research. (2025). Clinical Trial Cost Impacts of Supply Chain Restructuring Away from Countries of Concern.
- From Scientific Collaboration to Regulatory Chill: How the FDA Ban Tears Apart China-U.S. Biomedical Cooperation?

